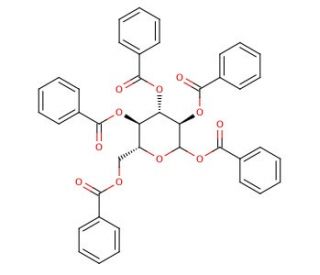
1,2,3,4,6-Penta-O-benzoyl-D-glucopyranoside (CAS 3006-49-3)
QUICK LINKS
1,2,3,4,6-Penta-O-benzoyl-D-glucopyranoside, a derivative of glucose, has been extensively utilized in research for its role in carbohydrate chemistry and as a versatile building block for synthesizing complex carbohydrates and glycoconjugates. Its chemical structure, featuring multiple benzoyl groups, facilitates selective deprotection reactions to generate a wide range of functionalized sugar derivatives. In carbohydrate chemistry, this compound serves as a valuable starting material for the synthesis of various glycosides, glycosyl donors, and acceptors through glycosylation reactions. Furthermore, 1,2,3,4,6-Penta-O-benzoyl-D-glucopyranoside is employed in the enzymatic synthesis of oligosaccharides and glycopeptides, where it acts as a glycosyl donor or acceptor to control the regioselectivity and stereochemistry of glycosylation reactions. Moreover, its ability to undergo chemoselective transformations enables the preparation of structurally diverse carbohydrate derivatives with tailored properties, such as solubility, stability, and bioactivity, making it invaluable in the synthesis of carbohydrate-based materials and glycoconjugates for applications in glycobiology, chemical biology, and materials science. Ongoing research continues to explore novel synthetic methodologies and applications of 1,2,3,4,6-Penta-O-benzoyl-D-glucopyranoside in advancing carbohydrate chemistry and glycobiology.
1,2,3,4,6-Penta-O-benzoyl-D-glucopyranoside (CAS 3006-49-3) References
- A new, efficient glycosylation method for oligosaccharide synthesis under neutral conditions: preparation and use of new DISAL donors. | Petersen, L. and Jensen, KJ. 2001. J Org Chem. 66: 6268-75. PMID: 11559173
- 1,2-Trans-selective synthesis of glycosyl boranophosphates and their utility as building blocks for the synthesis of phosphodiester-linked disaccharides. | Sato, K., et al. 2010. J Org Chem. 75: 2147-56. PMID: 20180549
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,3,4,6-Penta-O-benzoyl-D-glucopyranoside, 2.5 g | sc-397481 | 2.5 g | $320.00 |
