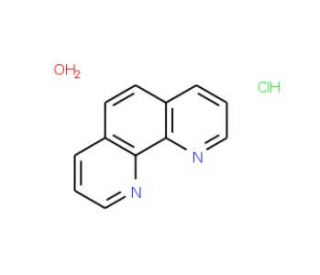
1,10-Phenanthroline Hydrochloride Monohydrate (CAS 18851-33-7)
QUICK LINKS
1,10-Phenanthroline Hydrochloride Monohydrate, a derivative of the aromatic compound phenanthroline, has been a focal point in scientific research, particularly in the fields of coordination chemistry, analytical chemistry, and materials science. Researchers have explored its versatile properties and unique structural features to explain its role in various chemical processes and applications. In coordination chemistry, 1,10-Phenanthroline Hydrochloride Monohydrate serves as a versatile ligand capable of forming stable complexes with transition metal ions. Its bidentate nature allows for the formation of chelate complexes, which exhibit distinctive coordination geometries and spectroscopic properties. Scientists have utilized these complexes in the design of catalysts, sensors, and molecular probes for investigating metal-ligand interactions and catalytic processes. Furthermore, 1,10-Phenanthroline Hydrochloride Monohydrate has been employed in analytical chemistry as a chelating agent for the determination of metal ions in complex matrices. Its selective complexation with specific metal ions enables sensitive detection and quantification using various spectroscopic and chromatographic techniques. Researchers have developed analytical methods based on 1,10-Phenanthroline Hydrochloride Monohydrate for environmental monitoring, pharmaceutical analysis, and forensic investigations.
1,10-Phenanthroline Hydrochloride Monohydrate (CAS 18851-33-7) References
- The expression and relaxant effect of bitter taste receptors in human bronchi. | Grassin-Delyle, S., et al. 2013. Respir Res. 14: 134. PMID: 24266887
- pH-dependent cross-linking of catechols through oxidation via Fe3+ and potential implications for mussel adhesion. | Fullenkamp, DE., et al. 2014. RSC Adv. 4: 25127-25134. PMID: 25243062
- Markedly Enhanced Surface Hydroxyl Groups of TiO₂ Nanoparticles with Superior Water-Dispersibility for Photocatalysis. | Wu, CY., et al. 2017. Materials (Basel). 10: PMID: 28772926
- New Insights into Solid Form Stability and Hydrate Formation: o-Phenanthroline HCl and Neocuproine HCl. | Braun, DE., et al. 2017. Molecules. 22: PMID: 29244765
- Functional and Pharmacological Analyses of the Role of Penicillium digitatum Proteases on Virulence. | Ballester, AR., et al. 2019. Microorganisms. 7: PMID: 31336863
- Bitter Taste Receptors (TAS2Rs) in Human Lung Macrophages: Receptor Expression and Inhibitory Effects of TAS2R Agonists. | Grassin-Delyle, S., et al. 2019. Front Physiol. 10: 1267. PMID: 31632299
- Characterization of an active ingredient made of nanoscale iron(oxyhydr)oxide for the treatment of hyperphosphatemia. | Bäumler, M., et al. 2021. RSC Adv. 11: 17669-17682. PMID: 35480163
- Process development for pilot-scale spray drying of ultrasmall iron (oxyhydr) oxide nanoparticles | Spicher, M. T.Schwaminger, S. P. and von der Haar-Leistl, D.Fellenberg, E.Berensmeier, S. 2024. Powder Technology. 433: 119186.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,10-Phenanthroline Hydrochloride Monohydrate, 25 g | sc-287209 | 25 g | $145.00 |
