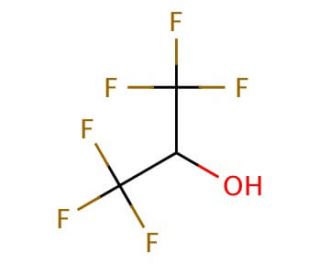
1,1,1,3,3,3-Hexafluoro-2-propanol (CAS 920-66-1)
See product citations (1)
QUICK LINKS
1,1,1,3,3,3-Hexafluoro-2-propanol (HFP) is a solution-phase peptide chemistry solvent. This fluorinated solvent enhances the efficiency of rhodium(I)-catalyzed [4+2] intramolecular cycloaddition of ether-tethered alkynyl dienes and [5+2] cycloaddition of alkynyl vinylcyclopropanes. It has a low vapor pressure, which makes it an ideal solvent for reactions that require low temperature and/or low pressure conditions. 1,1,1,3,3,3-Hexafluoro-2-propanol is used in a variety of scientific research applications, such as in the synthesis of fluorinated polymers, as a reagent in organic synthesis, and as a nonpolar solvent for a variety of reaction processes. It is also utilized in the development of novel catalysts and in the production of coatings and lubricants.
1,1,1,3,3,3-Hexafluoro-2-propanol (CAS 920-66-1) References
- Structure and solvation of melittin in 1,1,1,3,3,3-hexafluoro-2-propanol/water. | Gerig, JT. 2004. Biophys J. 86: 3166-75. PMID: 15111429
- Cationic Rh(I) catalyst in fluorinated alcohol: mild intramolecular cycloaddition reactions of ester-tethered unsaturated compounds. | Saito, A., et al. 2006. J Org Chem. 71: 6437-43. PMID: 16901128
- Solubility and supersaturation-dependent protein misfolding revealed by ultrasonication. | Lin, Y., et al. 2014. Langmuir. 30: 1845-54. PMID: 24059752
- Cα-H carries information of a hydrogen bond involving the geminal hydroxyl group: a case study with a hydrogen-bonded complex of 1,1,1,3,3,3-hexafluoro-2-propanol and tertiary amines. | Pal, U., et al. 2014. J Phys Chem A. 118: 1024-30. PMID: 24446840
- 1,1,1,3,3,3-Hexafluoro-2-propanol and 2,2,2-trifluoroethanol solvents induce self-assembly with different surface morphology in an aromatic dipeptide. | Reddy, SM., et al. 2014. Org Biomol Chem. 12: 6181-9. PMID: 24999600
- 1,1,1,3,3,3-Hexafluoro-2-propanol (HFIP) as a novel and effective solvent to facilely prepare cyclodextrin-assembled materials. | Kida, T., et al. 2014. Chem Commun (Camb). 50: 14245-8. PMID: 25284360
- Improved Schmidt Conversion of Aldehydes to Nitriles Using Azidotrimethylsilane in 1,1,1,3,3,3-Hexafluoro-2-propanol. | Motiwala, HF., et al. 2015. Molecules. 21: E45. PMID: 26729081
- Nuclear magnetic resonance evidence for the dimer formation of beta amyloid peptide 1-42 in 1,1,1,3,3,3-hexafluoro-2-propanol. | Shigemitsu, Y., et al. 2016. Anal Biochem. 498: 59-67. PMID: 26772162
- Observation of Intermolecular Hydrogen Bonding Interactions in Biosynthesized and Biodegradable Poly [(R)-3-hydroxybutyrate- co-(R)-3-hydroxyhexanoate] in Chloroform and 1,1,1,3,3,3-Hexafluoro-2-propanol (HFIP). | Sobieski, BJ., et al. 2017. Appl Spectrosc. 71: 2339-2343. PMID: 28441032
- Active Molybdenum-Based Anode for Dehydrogenative Coupling Reactions. | Beil, SB., et al. 2018. Angew Chem Int Ed Engl. 57: 2450-2454. PMID: 29318724
- Volatile Anesthetic Sevoflurane Precursor 1,1,1,3,3,3-Hexafluoro-2-Propanol (HFIP) Exerts an Anti-Prion Activity in Prion-Infected Culture Cells. | Shimizu, T., et al. 2021. Neurochem Res. 46: 2056-2065. PMID: 34043140
- Higher-Energy Hexafluoroisopropanol···Water Isomer and Its Large Amplitude Motions: Rotational Spectra and DFT Calculations. | Wu, B., et al. 2021. J Phys Chem A. 125: 10401-10409. PMID: 34846154
- 1,1,1,3,3,3-Hexafluoro-2-propanol (HFIP)-Assisted Catalyst-Free Sulfonation of Allylic Alcohols with Sulfinyl Amides. | Luo, Z., et al. 2022. Org Lett. 24: 741-745. PMID: 34989575
- Experimental and Theoretical Investigation of an Azaoxyallyl Cation-Templated Intramolecular Aryl Amination Leading to Oxindole Derivatives. | Bera, T., et al. 2022. Chemistry. 28: e202201208. PMID: 35943863
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,1,1,3,3,3-Hexafluoro-2-propanol, 25 ml | sc-251540 | 25 ml | $187.00 | |||
1,1,1,3,3,3-Hexafluoro-2-propanol, 100 ml | sc-251540A | 100 ml | $583.00 | |||
1,1,1,3,3,3-Hexafluoro-2-propanol, 500 ml | sc-251540B | 500 ml | $2299.00 | |||
1,1,1,3,3,3-Hexafluoro-2-propanol, 1 L | sc-251540C | 1 L | $3548.00 |
