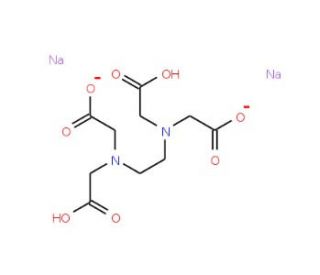
0.5M Sodium EDTA
See product citations (1)
QUICK LINKS
0.5M Sodium EDTA is a chemical solution commonly used in molecular biology and biochemical research for its ability to chelate divalent metal ions, particularly calcium (Ca²⁺) and magnesium (Mg²⁺). Ethylenediaminetetraacetic acid (EDTA) is a chelating agent that binds to metal ions, forming stable, water-soluble complexes which effectively remove these ions from the solution. This mechanism is crucial because divalent metal ions often serve as essential cofactors for many enzymatic activities, including those of DNases and RNases, which degrade DNA and RNA respectively. By chelating these metal ions, Sodium EDTA inhibits the activity of these nucleases, thus protecting nucleic acids from enzymatic degradation during procedures such as DNA and RNA extraction. Furthermore, in the context of research, the use of 0.5M Sodium EDTA extends to applications in cell biology where the removal of calcium ions disrupts cell membranes, aiding in cell lysis and the subsequent analysis of intracellular components. This solution is also pivotal in buffer systems used for gel electrophoresis of DNA, where it contributes to maintaining the integrity of the nucleic acids during the separation process. The ability of Sodium EDTA to bind metal ions also finds utility in water treatment processes in laboratories to prevent metal ion contamination and scale formation, underscoring its versatility and importance in scientific research.
0.5M Sodium EDTA References
- Final report on the safety assessment of EDTA, calcium disodium EDTA, diammonium EDTA, dipotassium EDTA, disodium EDTA, TEA-EDTA, tetrasodium EDTA, tripotassium EDTA, trisodium EDTA, HEDTA, and trisodium HEDTA. | Lanigan, RS. and Yamarik, TA. 2002. Int J Toxicol. 21 Suppl 2: 95-142. PMID: 12396676
- Quantification of protein in brain tissue by western immunoblot analysis. | Konradi, CL. 2003. Methods Mol Med. 79: 263-71. PMID: 12506702
- Some further observations on the interaction of EDTA with the myosin-ATP system. | FRIESS, EG., et al. 1954. Arch Biochem Biophys. 53: 311-3. PMID: 13208306
- Bismuth EDTA complex. | SAPEIKA, N. 1955. S Afr Med J. 29: 137-9. PMID: 14358876
- The inhibition of hematopoietic action of cobalt by ethylenediamine. tetraacetic acid (EDTA). | CHILD, GP. 1951. Science. 114: 466-7. PMID: 14892768
- A modified protocol for rapid DNA isolation from plant tissues using cetyltrimethylammonium bromide. | Allen, GC., et al. 2006. Nat Protoc. 1: 2320-5. PMID: 17406474
- Electrophoretic karyotypes of Torulopsis glabrata. | Kaufmann, CS. and Merz, WG. 1989. J Clin Microbiol. 27: 2165-8. PMID: 2584368
- Variation of electrophoretic karyotypes among clinical isolates of Candida albicans. | Merz, WG., et al. 1988. J Clin Microbiol. 26: 842-5. PMID: 3290238
- Enzymatic breakage and joining of deoxyribonucleic acid, I. Repair of single-strand breaks in DNA by an enzyme system from Escherichia coli infected with T4 bacteriophage. | Weiss, B. and Richardson, CC. 1967. Proc Natl Acad Sci U S A. 57: 1021-8. PMID: 5340583
- Determining EDTA in blood. | Sheppard, RL. and Henion, J. 1997. Anal Chem. 69: 477A-480A. PMID: 9253241
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
0.5M Sodium EDTA, 1 L | sc-286969 | 1 L | $21.00 |
