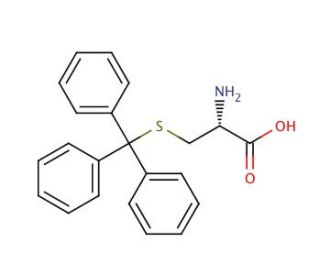
Molecular structure of S-Trityl-L-cysteine, CAS Number: 2799-07-7
S-Trityl-L-cysteine (CAS 2799-07-7)
See product citations (6)
Application:
S-Trityl-L-cysteine is a cell-permeable selective inhibitor of mitotic kinesin Eg5 and ATPase activities
CAS Number:
2799-07-7
Purity:
>98%
Molecular Weight:
363.5
Molecular Formula:
C22H21NO2S
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
S-Trityl-L-cysteine is a cell-permeable cysteine thioether compound that exhibits antitumor and antimitotic properties by inhibiting mitotic kinesin Eg5. S-Trityl-L-cysteine has been found to potently block that basal and microtubule-activated mitotic spindle kinesin ATPase activities. S-Trityl-L-cysteine may act to be 36-fold more potent than Monastrol (sc-202710) for inducing mitotic arrest in HeLa cells.
S-Trityl-L-cysteine (CAS 2799-07-7) References
- In vitro screening for inhibitors of the human mitotic kinesin Eg5 with antimitotic and antitumor activities. | DeBonis, S., et al. 2004. Mol Cancer Ther. 3: 1079-90. PMID: 15367702
- Identification of the protein binding region of S-trityl-L-cysteine, a new potent inhibitor of the mitotic kinesin Eg5. | Brier, S., et al. 2004. Biochemistry. 43: 13072-82. PMID: 15476401
- S-trityl-L-cysteine is a reversible, tight binding inhibitor of the human kinesin Eg5 that specifically blocks mitotic progression. | Skoufias, DA., et al. 2006. J Biol Chem. 281: 17559-69. PMID: 16507573
- Proteome analysis of apoptosis signaling by S-trityl-L-cysteine, a potent reversible inhibitor of human mitotic kinesin Eg5. | Kozielski, F., et al. 2008. Proteomics. 8: 289-300. PMID: 18186019
- Structure-activity relationship of S-trityl-L-cysteine analogues as inhibitors of the human mitotic kinesin Eg5. | Debonis, S., et al. 2008. J Med Chem. 51: 1115-25. PMID: 18266314
- Biochemical analysis of cellular target of S-trityl-L-cysteine derivatives using affinity matrix. | Shimizu, M., et al. 2010. Bioorg Med Chem Lett. 20: 1578-80. PMID: 20138511
- Docetaxel-resistant prostate cancer cells remain sensitive to S-trityl-L-cysteine-mediated Eg5 inhibition. | Wiltshire, C., et al. 2010. Mol Cancer Ther. 9: 1730-9. PMID: 20515952
- S-trityl-L-cysteine derivative induces caspase-independent cell death in K562 human chronic myeloid leukemia cell line. | Shimizu, M., et al. 2010. Cancer Lett. 298: 99-106. PMID: 20619960
- Inhibition of hepatitis C virus NS5B polymerase by S-trityl-L-cysteine derivatives. | Nichols, DB., et al. 2012. Eur J Med Chem. 49: 191-9. PMID: 22280819
- Doing the methylene shuffle--further insights into the inhibition of mitotic kinesin Eg5 with S-trityl L-cysteine. | Abualhasan, MN., et al. 2012. Eur J Med Chem. 54: 483-98. PMID: 22749640
- Optimized S-trityl-L-cysteine-based inhibitors of kinesin spindle protein with potent in vivo antitumor activity in lung cancer xenograft models. | Good, JA., et al. 2013. J Med Chem. 56: 1878-93. PMID: 23394180
- Photocontrol of the mitotic kinesin Eg5 using a novel S-trityl-L-cysteine analogue as a photochromic inhibitor. | Ishikawa, K., et al. 2014. J Biochem. 155: 257-63. PMID: 24451491
- S-trityl-L-cysteine, a novel Eg5 inhibitor, is a potent chemotherapeutic strategy in neuroblastoma. | Wu, W., et al. 2018. Oncol Lett. 16: 1023-1030. PMID: 29963178
- Antiproliferative S-Trityl-l-Cysteine -Derived Compounds as SIRT2 Inhibitors: Repurposing and Solubility Enhancement. | Radwan, MO., et al. 2019. Molecules. 24: PMID: 31510043
- Design, synthesis, and evaluation of a novel prodrug, a S-trityl-l-cysteine derivative targeting kinesin spindle protein. | Fukai, R., et al. 2021. Eur J Med Chem. 215: 113288. PMID: 33640763
Inhibitor of:
2610039C10Rik, 4930404A10Rik, 4930534B04Rik, 5830418K08Rik, 9830001H06Rik, ACYP2, AK129341, ANKRD56, ASPM, ATPase, AUNIP, C16orf63_0610037P05Rik, C17orf55, C1orf96, C20orf19, CASC5, CCDC38, CCDC52, Cdc16, Cdc4, CENP (Centromere protein), CENP-C, CENP-F, CENP-O, CENP-T, CENPBD1, CENPVL3, centlein, Centrobin, CEP152, CKAP5, CSPP1, CTAGE1, CTAGE2, CUG2, D14Ertd500e, EG244495, Eg5, EML6, Esp1, FAM44B, fzr, hCAP-G, hCAP-H, HYLS1, INSC, Katanin p60 A1, KIAA1009, KIF (Kinesin family), KIF20A, KIF6, Kinesin, kinesin light chain, KLC, Klp3, KNL2, KRCC1, LRRIQ1, MACF1, Mad 3, MAD2L1BP, MAP-7, MAPRE (microtubule associated protein RP/EB), MDM1, MEIKIN, MFAP1B, mKIAA1853, MKLP-1, neuronal kinesin heavy chain, NuMA, OIP5, POC5, PRR19, RCC1, rotatin, SAP 114, scc-112, Sgo2, Sil, Ska2, SPANX-N1, TNP2, ubiquitous kinesin heavy chain, and WDR51B.Activator of:
β8 Tubulin, 1700001P01Rik, APC4, APC7, ASPM, Aurora A, AW555464, C14orf106, C19orf21, Cdc11, CENP-M, CENP-N, CENP-O, CENP-T, CENP-V, CEP63, CKAP2L, CLASP2, CSAP, DNAL1, DSN1, EG668210, FAM110A, FAM154A, Gm1305, HAUS3, HAUS6, hCAP-D2, Hec1, KIAA0895L_4931428F04Rik, KIF4A, KIFC2, MZT1, NSL1, SEL-10, Ska1, Spc25, SPDL-1, Speriolin, TACC1, TACC2, TTBK1, TTK_Esk, UIP1, WDR59, and ZWINT.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
S-Trityl-L-cysteine, 1 g | sc-202799 | 1 g | $32.00 | |||
S-Trityl-L-cysteine, 5 g | sc-202799A | 5 g | $66.00 |
