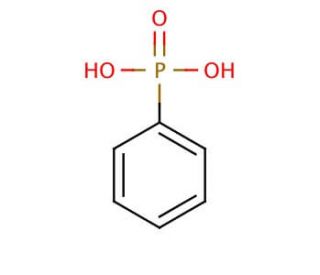
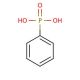
Phenylphosphonic acid (CAS 1571-33-1)
QUICK LINKS
Phenylphosphonic acid (PPA) is a versatile organic compound extensively employed in diverse scientific research applications. Its broad spectrum of uses includes serving as a catalyst in various chemical syntheses and participating in biochemical reactions. Moreover, Phenylphosphonic acid finds application as a non-ionic surfactant in laboratory experiments. As a catalyst, Phenylphosphonic acid plays a pivotal role in facilitating biochemical reactions such as the synthesis of peptides and oligonucleotide analogs. It also proves indispensable in synthesizing a plethora of organic compounds, encompassing polymers and dyes. The secret behind its catalytic prowess lies in its ability to form hydrogen bonds with other molecules, fostering the creation of new bonds between them. Furthermore, Phenylphosphonic acid exhibits the fascinating capacity to form complexes with metal ions, unleashing their potential as catalysts for various reactions. Phenylphosphonic acid stands as an invaluable tool in the arsenal of scientific research, elevating the possibilities of chemical synthesis and biochemical exploration. With its multifaceted applications and intriguing mechanism of action, the future holds exciting promise for further advances in harnessing the potential of Phenylphosphonic acid.
Phenylphosphonic acid (CAS 1571-33-1) References
- Phosphate-imprinted magnetic nanoparticles using phenylphosphonic acid as a template for excellent recognition of tyrosine phosphopeptides. | Liu, Q., et al. 2018. Talanta. 186: 346-353. PMID: 29784371
- Protolysis and Complex Formation of Organophosphorus Compounds-Characterization by NMR-Controlled Titrations. | Hägele, G. 2019. Molecules. 24: PMID: 31492010
- Solvent-Free Synthesis of Nano Zirconium Phenylphosphonates with Molten Phenylphosphonic Acid. | Contreras-Ramirez, A., et al. 2020. Chemistry. 26: 6185-6194. PMID: 32128881
- Semiconductive microporous hydrogen-bonded organophosphonic acid frameworks. | Tholen, P., et al. 2020. Nat Commun. 11: 3180. PMID: 32576877
- Analysis of metabolites and metabolic pathways in three maize (Zea mays L.) varieties from the same origin using GC-MS. | Zhang, L., et al. 2020. Sci Rep. 10: 17990. PMID: 33093508
- Organic modification of layered zirconium phosphate/phosphonate for controlled release of therapeutic inorganic ions. | Nakamura, J., et al. 2021. Sci Technol Adv Mater. 22: 1000-1012. PMID: 34924816
- Multi-functional porous organic polymers for highly-efficient solid-phase extraction of β-agonists and β-blockers in milk. | Wu, C., et al. 2021. RSC Adv. 11: 28925-28933. PMID: 35478535
- Evolution of Artificial Arginine Analogues-Fluorescent Guanidiniocarbonyl-Indoles as Efficient Oxo-Anion Binders. | Sebena, D., et al. 2022. Molecules. 27: PMID: 35566361
- Phosphonic acid-containing inhibitors of tyrosyl-DNA phosphodiesterase 1. | Zhao, XZ., et al. 2022. Front Chem. 10: 910953. PMID: 36051621
- Phenylphosphonic acid as a grain-refinement additive for a stable lithium metal anode. | Zou, P., et al. 2022. Chem Commun (Camb). 58: 12724-12727. PMID: 36314285
- Self-Assembly of Cyclodextrin-Coated Nanoparticles:Fabrication of Functional Nanostructures for Sensing and Delivery. | Cengiz, B., et al. 2023. Molecules. 28: PMID: 36770743
- Investigation on the Photodegradation Stability of Acrylic Acid-Grafted Poly(butylene carbonate-co-terephthalate)/Organically Modified Layered Zinc Phenylphosphonate Composites. | Lee, YF. and Wu, TM. 2023. Polymers (Basel). 15: PMID: 36904517
- Synthesis of FeOOH-Loaded Aminated Polyacrylonitrile Fiber for Simultaneous Removal of Phenylphosphonic Acid and Phosphate from Aqueous Solution. | Zhou, R., et al. 2023. Polymers (Basel). 15: PMID: 37112065
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Phenylphosphonic acid, 5 g | sc-236378 | 5 g | $28.00 | |||
Phenylphosphonic acid, 100 g | sc-236378A | 100 g | $31.00 |
