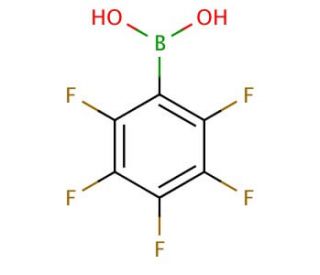
Pentafluorophenylboronic acid (CAS 1582-24-7)
QUICK LINKS
Pentafluorophenylboronic acid is a compound that functions as a versatile building block in organic synthesis. Its mechanism of action involves its ability to form stable complexes with diols and polyols, making it a useful tool for the formation of carbon-carbon and carbon-heteroatom bonds. Pentafluorophenylboronic acid may act as a Lewis acid, facilitating the activation of substrates and promoting various cross-coupling reactions. Pentafluorophenylboronic acid′s mode of action involves its interaction with nucleophiles, enabling the formation of arylboronates and subsequent transformations. Pentafluorophenylboronic acid may play role in the development of new materials and biochemicals, as well as in the synthesis of complex organic molecules. Its mechanism of action at the molecular level involves its ability to participate in Suzuki-Miyaura coupling reactions, enabling the construction of biaryl compounds and other valuable organic products.
Pentafluorophenylboronic acid (CAS 1582-24-7) References
- Suzuki-Miyaura coupling reaction using pentafluorophenylboronic acid. | Korenaga, T., et al. 2005. Org Lett. 7: 4915-7. PMID: 16235921
- Mild and general conditions for the cross-coupling of aryl halides with pentafluorobenzene and other perfluoroaromatics. | Lafrance, M., et al. 2006. Org Lett. 8: 5097-100. PMID: 17048852
- An experimental and theoretical study of molecular structure and vibrational spectra of 3- and 4-pyridineboronic acid molecules by density functional theory calculations. | Kurt, M., et al. 2008. Spectrochim Acta A Mol Biomol Spectrosc. 70: 664-73. PMID: 17933583
- 9,10-Disubstituted octafluoroanthracene derivatives via palladium-catalyzed cross-coupling. | Tannaci, JF., et al. 2008. J Org Chem. 73: 7895-900. PMID: 18788782
- Boronic Acid catalyzed friedel-crafts reactions of allylic alcohols with electron-rich arenes and heteroarenes. | McCubbin, JA., et al. 2010. J Org Chem. 75: 959-62. PMID: 20055455
- Heterotriangulenes π-expanded at bridging positions. | Chou, CM., et al. 2014. Org Lett. 16: 2868-71. PMID: 24851675
- Molecular design of cage iron(II) and cobalt(II,III) complexes with a second fluorine-enriched superhydrophobic shell. | Belov, AS., et al. 2015. Dalton Trans. 44: 3773-84. PMID: 25607531
- A surprising substituent effect provides a superior boronic acid catalyst for mild and metal-free direct Friedel-Crafts alkylations and prenylations of neutral arenes. | Ricardo, CL., et al. 2015. Chemistry. 21: 4218-23. PMID: 25678266
- Boronic acid/Brønsted acid co-catalyst systems for the synthesis of 2H-chromenes from phenols and α,β-unsaturated carbonyls. | Dimakos, V., et al. 2016. Org Biomol Chem. 14: 6703-11. PMID: 27314604
- Gold-Catalyzed Direct Oxidative Arylation with Boron Coupling Partners. | Hofer, M., et al. 2017. Angew Chem Int Ed Engl. 56: 1021-1025. PMID: 28000976
- Base-Catalyzed Aryl-B(OH)2 Protodeboronation Revisited: From Concerted Proton Transfer to Liberation of a Transient Aryl Anion. | Cox, PA., et al. 2017. J Am Chem Soc. 139: 13156-13165. PMID: 28823150
- Aryl Boronic Acid Catalysed Dehydrative Substitution of Benzylic Alcohols for C-O Bond Formation. | Estopiñá-Durán, S., et al. 2019. Chemistry. 25: 3950-3956. PMID: 30629761
- Perfluorophenylboronic acid-catalyzed direct α-stereoselective synthesis of 2-deoxygalactosides from deactivated peracetylated d-galactal. | Tatina, MB., et al. 2019. Chem Commun (Camb). 55: 12204-12207. PMID: 31549691
- Arylboronic Acid Catalyzed C-Alkylation and Allylation Reactions Using Benzylic Alcohols. | Estopiñá-Durán, S., et al. 2020. Org Lett. 22: 7547-7551. PMID: 32959662
- Armor-like Inorganic-rich Cathode Electrolyte Interphase Enabled by the Pentafluorophenylboronic Acid Additive for High-voltage Li||NCM622 Batteries. | Yang, Y., et al. 2023. Angew Chem Int Ed Engl. 62: e202300057. PMID: 36929622
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pentafluorophenylboronic acid, 5 g | sc-250666 | 5 g | $84.00 |
