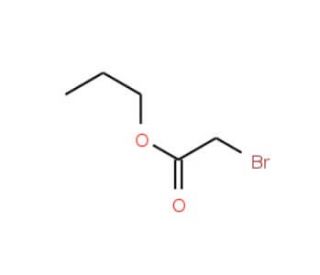
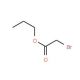
n-Propyl bromoacetate (CAS 35223-80-4)
QUICK LINKS
n-Propyl bromoacetate is a versatile chemical compound extensively employed in numerous industrial and scientific applications. This flammable, colorless liquid emits a strong odor. n-Propyl bromoacetate possesses a range of unique properties that render it valuable in a wide variety of applications. n-Propyl bromoacetate finds widespread use in scientific research, particularly in the fields of biochemistry and molecular biology. Notably, n-Propyl bromoacetate is employed in generating fluorescent labels for proteins and other molecules, enabling the study of their structure and function. Acting as a reagent in chemical reactions, n-Propyl bromoacetate facilitates the breaking and formation of bonds between molecules. It also functions as a catalyst in the conversion of inorganic compounds into organic compounds. Additionally, n-Propyl bromoacetate demonstrates catalytic properties in the formation of peptides and other biologically active compounds.
n-Propyl bromoacetate (CAS 35223-80-4) References
- Monoester copillar[5]arenes: synthesis, unusual self-inclusion behavior, and molecular recognition. | Chen, Y., et al. 2013. Chemistry. 19: 7064-70. PMID: 23568621
- Determination of trace level genotoxic impurities in small molecule drug substances using conventional headspace gas chromatography with contemporary ionic liquid diluents and electron capture detection. | Ho, TD., et al. 2014. J Chromatogr A. 1361: 217-28. PMID: 25151038
- Atomic physicochemical parameters for three-dimensional-structure-directed quantitative structure-activity relationships. 2. Modeling dispersive and hydrophobic interactions. | Ghose, AK. and Crippen, GM. 1987. J Chem Inf Comput Sci. 27: 21-35. PMID: 3558506
- Protein Release by Controlled Desorption from Transiently Cationic Nanoparticles. | Cheung, TH., et al. 2023. ACS Appl Mater Interfaces.. PMID: 36703567
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
n-Propyl bromoacetate, 5 g | sc-269888 | 5 g | $65.00 |
