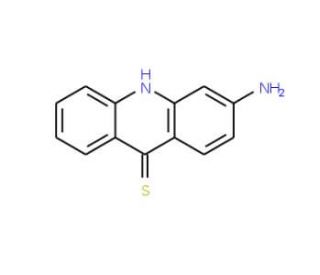
3-ATA (CAS 129821-08-5)
See product citations (2)
QUICK LINKS
3-ATA, known chemically as 3-amino-1,2,4-triazole and identified by CAS number 129821-08-5, is a triazole derivative that functions as a potent inhibitor of the enzyme catalase. Catalase is an essential enzyme that catalyzes the decomposition of hydrogen peroxide into water and oxygen, serving as a critical protective mechanism against oxidative stress in cells. By inhibiting catalase, 3-ATA increases the levels of hydrogen peroxide within cells. This rise in hydrogen peroxide can induce oxidative stress, which is useful for research purposes in studying the effects of reactive oxygen species (ROS) on cellular processes and the mechanisms cells employ to contend with oxidative damage. In research settings, 3-ATA is utilized to examine the cellular responses to increased oxidative stress, including the activation of antioxidant defense pathways, the role of ROS in regulating cell signaling pathways, and the impact of oxidative stress on cell viability and function. Additionally, its role in influencing oxidative balance makes 3-ATA a valuable tool for investigating the physiological and pathological roles of catalase and other antioxidative enzymes. Through these applications, 3-ATA contributes to a deeper understanding of redox biology and the complex network of cellular responses to oxidative challenges.
3-ATA (CAS 129821-08-5) References
- [Electron microscopic studies on primary culture cells from human oral tissues. II. Ultrastructure of the epithelial cells from squamous cell carcinoma in oral cavity (author's transl)]. | Yuki, K. 1975. Kokubyo Gakkai Zasshi. 42: 444-72. PMID: 1063237
- The osmoregulatory and the amino acid-regulated responses of system A are mediated by different signal transduction pathways. | López-Fontanals, M., et al. 2003. J Gen Physiol. 122: 5-16. PMID: 12810851
- 3-Amino thioacridone, a selective cyclin-dependent kinase 4 inhibitor, attenuates kainic acid-induced apoptosis in neurons. | Verdaguer, E., et al. 2003. Neuroscience. 120: 599-603. PMID: 12895500
- Roles of cyclin-dependent kinase 4 and p53 in neuronal cell death induced by doxorubicin on cerebellar granule neurons in mouse. | Otsuka, Y., et al. 2004. Neurosci Lett. 365: 180-5. PMID: 15246544
- New acridone inhibitors of human herpes virus replication. | Bastow, KF. 2004. Curr Drug Targets Infect Disord. 4: 323-30. PMID: 15578973
- 3-amino thioacridone inhibits DNA synthesis and induce DNA damage in T-cell acute lymphoblastic leukemia (T-ALL) in a p16-dependent manner. | Diccianni, MB., et al. 2004. J Exp Ther Oncol. 4: 223-37. PMID: 15724842
- Acridine and acridone derivatives, anticancer properties and synthetic methods: where are we now? | Belmont, P., et al. 2007. Anticancer Agents Med Chem. 7: 139-69. PMID: 17348825
- Serum starvation induces G1 arrest through suppression of Skp2-CDK2 and CDK4 in SK-OV-3 cells. | Shin, JS., et al. 2008. Int J Oncol. 32: 435-9. PMID: 18202766
- CDK2 and CDK4 play important roles in promoting the proliferation of SKOV3 ovarian carcinoma cells induced by tumor-associated macrophages. | Yang, Y., et al. 2014. Oncol Rep. 31: 2759-68. PMID: 24700371
- A comprehensive review on acridone based derivatives as future anti-cancer agents and their structure activity relationships. | Yadav, TT., et al. 2022. Eur J Med Chem. 239: 114527. PMID: 35717872
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-ATA, 5 mg | sc-202414 | 5 mg | $327.00 |
