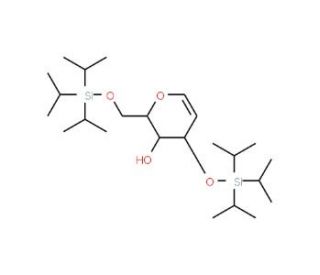
3,6-Di-O-(triisopropylsilyl)-D-glucal
QUICK LINKS
3,6-Di-O-(triisopropylsilyl)-D-glucal is a synthetic derivative of D-glucal featuring two triisopropylsilyl (TIPS) protecting groups. This compound is widely utilized in carbohydrate chemistry due to its functional groups, which provide protection for the hydroxyl groups at positions 3 and 6, ensuring selective reactions at other positions. The TIPS groups are bulky, providing steric hindrance, which improves selectivity during glycosylation reactions, making it easier to control the synthesis of specific glycosidic bonds. Researchers often use 3,6-Di-O-(triisopropylsilyl)-D-glucal in glycosylation reactions to synthesize complex oligosaccharides and glycoconjugates. The protected D-glucal can be converted into a glycosyl donor or glycal intermediate, facilitating the formation of glycosidic bonds with high stereoselectivity. Its structure is particularly advantageous for studying carbohydrate-protein interactions, allowing for the creation of oligosaccharide structures that mimic natural glycans in biological systems. The compound′s stability and selective reactivity enable researchers to investigate the influence of glycosylation patterns on biological functions and to map carbohydrate epitopes for antibody-antigen recognition studies. This model glucal compound serves as a vital tool in the development of advanced synthetic methodologies for carbohydrate chemistry and for expanding our understanding of glycan structure-activity relationships in biochemistry.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3,6-Di-O-(triisopropylsilyl)-D-glucal, 500 mg | sc-299165 | 500 mg | $304.00 |
