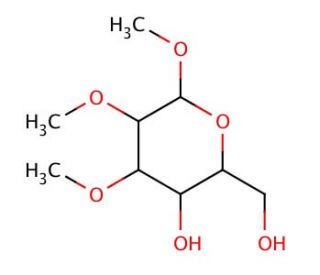
1,2,3-Tri-O-methyl-α-D-glucopyranose (CAS 14048-30-7)
QUICK LINKS
1,2,3-Tri-O-methyl-α-D-glucopyranose, a derivative of D-glucose, serves as a valuable tool in carbohydrate chemistry and glycobiology research. Its mechanism of action lies in its chemical structure, which consists of three methyl groups substituted at the hydroxyl positions of the glucose ring. This modification imparts unique properties to the molecule, making it resistant to enzymatic hydrolysis by glycosidases. Researchers utilize 1,2,3-Tri-O-methyl-α-D-glucopyranose as a building block in the synthesis of complex carbohydrates and glycoconjugates for studying glycan-protein interactions, carbohydrate-mediated signaling pathways, and glycan biosynthesis. Additionally, this compound finds applications in NMR spectroscopy studies for elucidating the three-dimensional structures of oligosaccharides and glycoconjugates, providing valuable insights into their conformational preferences and molecular recognition properties. Furthermore, 1,2,3-Tri-O-methyl-α-D-glucopyranose serves as a precursor in the preparation of per-O-methylated derivatives of carbohydrates, which are utilized as analytical standards and reference compounds in glycomics and glycoproteomics research. Through its chemical stability and versatile applications, 1,2,3-Tri-O-methyl-α-D-glucopyranose continues to advance our understanding of carbohydrate structure-function relationships and pave the way for innovative research approaches aimed at unraveling the roles of glycans in various biological processes.
1,2,3-Tri-O-methyl-α-D-glucopyranose (CAS 14048-30-7) References
- Asymmetry of the hexose transfer system in human erythrocytes. Experiments with non-transportable inhibitors. | Baker, GF., et al. 1978. J Physiol. 278: 377-88. PMID: 671317
- Asymmetry of hexose transfer system in erythrocytes of fetal and new-born guinea-pigs. | Aubby, DS. and Widdas, WF. 1980. J Physiol. 309: 317-27. PMID: 7252868
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,3-Tri-O-methyl-α-D-glucopyranose, 100 mg | sc-220533 | 100 mg | $300.00 |
