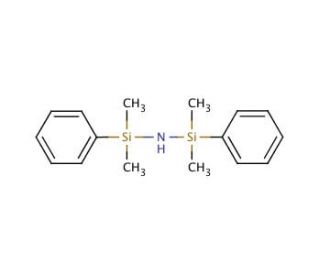
1,1,3,3-Tetramethyl-1,3-diphenyldisilazane (CAS 3449-26-1)
QUICK LINKS
1,1,3,3-Tetramethyl-1,3-diphenyldisilazane, known as DMTD, is an organosilicon compound with a wide range of scientific applications that span decades. It is a stable and colorless liquid, non-volatile and unable to dissolve in water, but readily soluble in various organic solvents. This versatile compound holds a prominent role as the most widely used silicon-based reagent in organic synthesis, having contributed significantly to the creation of diverse compounds. In the realm of scientific research, 1,1,3,3-Tetramethyl-1,3-diphenyldisilazane has proven invaluable in various fields such as organic synthesis and biochemistry. Its catalytic properties have played a vital role in facilitating the synthesis of numerous compounds. Moreover, its efficacy as a reagent has been instrumental in the creation of various organic compounds, including peptides and other biopolymers. Functioning as a nucleophilic reagent, 1,1,3,3-Tetramethyl-1,3-diphenyldisilazane finds its application in the Mitsunobu reaction. In this process, the nucleophilic reagent attacks the electrophilic carbon atom of an aromatic amine, giving rise to an intermediate product, which is subsequently hydrolyzed to yield the desired final product. This highly selective reaction has proved to be a powerful tool in synthesizing a diverse array of compounds with great precision.
1,1,3,3-Tetramethyl-1,3-diphenyldisilazane (CAS 3449-26-1) References
- Combined thin layer chromatography and gas chromatography with mass spectrometric analysis of lipid classes and fatty acids in malnourished polar bears (Ursus maritimus) which swam to Iceland. | Eibler, D., et al. 2017. J Chromatogr B Analyt Technol Biomed Life Sci. 1046: 138-146. PMID: 28183043
- Silazanes plus MCl4-substitution vs. rearrangement reactions | C. Lehnert, J. Wagler, E. Kroke & G. Roewer. 2006. Chemistry of Heterocyclic Compounds. 42: 1574–1584.
- A review of the R3Si–NH–SiR3–type disilazanes: From synthesis to applications | Krzysztof Kuciński, Hanna Stachowiak, Dariusz Lewandowski, Marcin Gruszczyński, Paulina Lampasiak, Grzegorz Hreczycho. 2022. Journal of Organometallic Chemistry. 961.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,1,3,3-Tetramethyl-1,3-diphenyldisilazane, 1 g | sc-237650 | 1 g | $31.00 |
