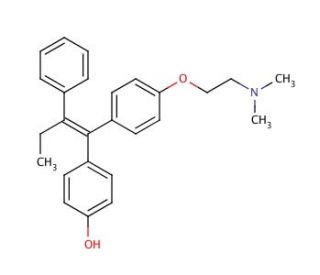
(Z)-4-Hydroxytamoxifen (CAS 68047-06-3)
See product citations (20)
QUICK LINKS
(Z)-4-Hydroxytamoxifen is an active metabolite of tamoxifen (sc-208414), an anti-estrogen agent. (Z)-4-Hydroxytamoxifen binds estrogen receptors and estrogen-related receptors with estrogenic and anti-estrogenic effects. (Z)-4-Hydroxytamoxifen is a cell permeable, selective estrogen receptor modulator. (Z)-4-Hydroxytamoxifen has been reported to irreversibly inhibit purified protein kinase C (PKC) in vitro through oxidative inactivation of the catalytic domain. Additionally, microsomal activation of 4-hydroxytamoxifen has been reported to lead to the formation of DNA adducts in vitro. The formation of these DNA adducts were also reported to be dependent on the cofactor utilized for microsomal activation. (Z)-4-Hydroxytamoxifen has also been shown to inhibit lipid peroxidation.
(Z)-4-Hydroxytamoxifen (CAS 68047-06-3) References
- Synthesis and sulfatase inhibitory activities of (E)- and (Z)-4-hydroxytamoxifen sulfamates. | Chu, GH., et al. 1999. Bioorg Med Chem Lett. 9: 141-4. PMID: 10021916
- New Highly Stereoselective Synthesis of (Z)-4-Hydroxytamoxifen and (Z)-4-Hydroxytoremifene via McMurry Reaction. | Gauthier, S., et al. 1996. J Org Chem. 61: 3890-3893. PMID: 11667248
- Synthesis of (Z)-4-hydroxytamoxifen and (Z)-2-[4-[1-(p-hydroxyphenyl)-2-phenyl]-1butenyl]phenoxyacetic acid. | Detsi, A., et al. 2002. J Org Chem. 67: 4608-11. PMID: 12076167
- Simple and efficient production of (Z)-4-hydroxytamoxifen, a potent estrogen receptor modulator. | Yu, DD. and Forman, BM. 2003. J Org Chem. 68: 9489-91. PMID: 14629178
- Design, synthesis, and biological evaluation of doxorubicin-formaldehyde conjugates targeted to breast cancer cells. | Burke, PJ. and Koch, TH. 2004. J Med Chem. 47: 1193-206. PMID: 14971899
- Activity levels of tamoxifen metabolites at the estrogen receptor and the impact of genetic polymorphisms of phase I and II enzymes on their concentration levels in plasma. | Mürdter, TE., et al. 2011. Clin Pharmacol Ther. 89: 708-17. PMID: 21451508
- Tamoxifen promotes superoxide production in platelets by activation of PI3-kinase and NADPH oxidase pathways. | Shah, VP., et al. 2012. Thromb Res. 129: 36-42. PMID: 21875743
- Evaluation of tamoxifen and metabolites by LC-MS/MS and HPLC methods. | Heath, DD., et al. 2014. Br J Biomed Sci. 71: 33-9. PMID: 24693573
- Association of CYP2C19*2 and associated haplotypes with lower norendoxifen concentrations in tamoxifen-treated Asian breast cancer patients. | Lim, JS., et al. 2016. Br J Clin Pharmacol. 81: 1142-52. PMID: 26799162
- The formation of estrogen-like tamoxifen metabolites and their influence on enzyme activity and gene expression of ADME genes. | Johänning, J., et al. 2018. Arch Toxicol. 92: 1099-1112. PMID: 29285606
- Heterodimeric GW7604 Derivatives: Modification of the Pharmacological Profile by Additional Interactions at the Coactivator Binding Site. | Knox, AK., et al. 2021. J Med Chem. 64: 5766-5786. PMID: 33904307
- Patient-derived scaffolds as a drug-testing platform for endocrine therapies in breast cancer. | Gustafsson, A., et al. 2021. Sci Rep. 11: 13334. PMID: 34172801
- Evaluation of mitochondrial oxidative toxicity in mammalian cardiomyocytes by determining the highly reproducible and reliable increase in mitochondrial superoxides after exposure to therapeutic drugs. | Ahn, D., et al. 2022. Toxicol In Vitro. 83: 105393. PMID: 35618243
- Tamoxifen modulates protein kinase C via oxidative stress in estrogen receptor-negative breast cancer cells. | Gundimeda, U., et al. 1996. J Biol Chem. 271: 13504-14. PMID: 8662863
- Production of 8-hydroxy-2'-deoxguanosine in DNA by microsomal activation of tamoxifen and 4-hydroxytamoxifen. | Ye, Q. and Bodell, WJ. 1996. Carcinogenesis. 17: 1747-50. PMID: 8761436
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
(Z)-4-Hydroxytamoxifen, 1 mg | sc-3542C | 1 mg | $220.00 | |||
(Z)-4-Hydroxytamoxifen, 5 mg | sc-3542 | 5 mg | $281.00 | |||
(Z)-4-Hydroxytamoxifen, 10 mg | sc-3542B | 10 mg | $408.00 | |||
(Z)-4-Hydroxytamoxifen, 25 mg | sc-3542A | 25 mg | $718.00 | |||
(Z)-4-Hydroxytamoxifen, 50 mg | sc-3542D | 50 mg | $1377.00 | |||
(Z)-4-Hydroxytamoxifen, 100 mg | sc-3542E | 100 mg | $2397.00 |
