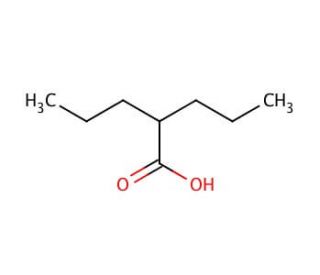
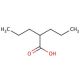
Activator of:
α-synuclein, β-Actin, β-catenin, β-synuclein, β1-Syntrophin, β2B Tubulin, δ-catenin, μ-protocadherin, 1110059E24Rik, 1700023E05Rik, 2410089E03Rik, 2900008C10Rik, 4930557A04Rik, 4931431F19Rik, 5330417C22Rik, 60 kDa Ro/SSA, A1-b, AACT, AADACL4, AANAT, ABAT, ABCF2, ABCG2, Ada1, ADAM5, ADAR2, advillin, AEBP2, AF9, AHI1, AI413782, AKNA, ALDH1A1, ALKBH7, alpha-L-iduronidase, ALS2CR11, ANKRD18A, ANKRD25, ANKRD34A, ANKS4B, AOF1, API5, APOBEC3B, ARALAR, AREBP, ARHGEF16, ARID1B, ARID3B, Armadillo (β-catenin), ArpM1, ARPP-21, ARX, ASCC1, ASCL1, ASCL2, ASCL5, ASGPR2, ASMT, ASTE1, Ataxin-1, Ataxin-10, ATF-7IP, Atg4a, atonal, ATP5 (Complex V; Mitochondrial ATP synthase), ATXN7L2, ATXN7L3, BAF, BAF170, BAF53B, BAF60a, BAFL, BAGE3, BAIAP2L1, Barhl1, Barhl2, BBS3, Bcl-11a, Bdf1, BDV, Bet1, Bex1, Bex2, Bex4, Bex6, Bhlhb9, BIG1, BIP, BLOS2, BM88, BMAL1, Bpifa6, BPTF, BRCA1, BRCAA1, BRD3, BRD8, BRINP2, Brn-2, BTBD8, BTF3, BUD13, C130074G19Rik, C14orf56, C16orf13_0610011F06Rik, C1orf105, C1orf112, C1orf146, C1orf181, C1orf227, C1orf54, C1orf57, C1orf65, C1orf87, C20orf54, C22orf42, C230097I24Rik, C230099D08Rik, C2orf49_AI597479, C330021F23Rik, C3orf1_4930455C21Rik, C3orf23, C3orf30, C3orf45, C3orf58, C6orf89, cadherin-28, CAF-1 p150, Calcipressin-2, Calcyon, casein kinase Iε, CASPR4, cbp146, CBX6, CBX7, CCDC21, CCDC40, CCP5, CD20, CD200R4, CD225, CD26, CD3, CD34, CD77 synthase (A4GALT), CDKL5, Cdx4, CDY2, CECR2, CENP-A, CENP-B, CENP-R, CENP-S, Cerebellin 3, CGI-69, CGRP2, CHD2, CHIP, Chr-A, CHRAC15, Chromodomain Helicase DNA binding protein (CHD), CHST5, Chx10, CIR, Ckt2, CLAMP, CLCA2_Clac2, CLDND1, CLEC-4F, CLIP-170_restin, Clock, CMV, CMV pp52, CMV US28, Cnbp2, CNTNAP3, Cntnap5c, COL6A6, connexin 33, connexin 45, Contactin 6, Corin, CRF, Cripto-3, CRMP-4, Cruz Tag 09TM, CrxOS, Csl, CSMD2, CSS2, CTLA-4, cubilin, CUEDC2, cylindromatosis 1, CYP11A1, CYP26A1, Cyp3a41b, CYP4A, cystatin 12, D3Ertd751e, D530049N12Rik, D930020B18Rik, Dapper1, Dapper2, DAT, Daxx, DBC1, DBX1, DCAMKL2, DCAMKL3, DcR2, DDA3, DDO, DDX17, DEDD, Delta-4, Deltex-4, dendrin, DGCR2, Dkk-3, DMRT3, Dmrtc1c, DNA pol ε, DNA pol ε cat, DNAH10, DNAH3, DNAHC12, DNAHC7L, DNLC2A, Dopey-2, DPF1, DPPA2, DPPA4, DPPA5, DPRP, DR1, DRD1a, Drebrin, DSCAML1, DSCR 1, DSCR 8, Duo, DUX, DUX1, DUX4, DUXBL, DYDC1, Dynein HC, Dynlt1d, dysferlin activators, dystrophin, DYX1C1, DZIP3, E130120F12Rik, E130309D14Rik, E130311K13Rik, E230019M04Rik, E230028L10Rik, E2A, EAF1, Ear10, Ear14, Ear3, Ear4, Ear7, EBV Bam HI Z, EBV EBNA-1, EBV EBNA-LP, EBV gp110 Envelope Protein, EBV gp220, EBV viral capsid antigen, EBV ZEBRA, EED, EF-CBP1, EFO2, EG210583, EG210853, EG236451, EG236663, EG240038, EG243866, EG384813, EG385328, EG434676, EG435601, EG436188, EG436523, EG546205, EG623186, EG626215, EG626305, EG626456, EG626575, EG626682, EG626740, EG626970, EG628359, EG669998, EGL-27, eHAND, EHZF, EID-1, eIF1AX, eIF3δ, emerin, Emx2, EN-1, ERVK6, ERVMER34-1, ESC, ESET, ETV3L, EVC, Evi-1, EVI2B, EZHIP, F830116E18Rik, FAM159A, FAM168A, FAM208A, FAM43B, FAM55C, FAM69A, FASP1, FBL11, FCP1, FEM-1, FEZF1, FGD3, Fhit, Fibrocystin L, FIV gp36, FIV p27, FKBP51, FLJ43505, FLJ44635, FMR1, FMR2, FNDC7, Fox-1, FOXB1, FOXI1, FOXL1, FOXN4, FOXP3, FRAT1, FRG2B, FRG2C, frizzled-3, frizzled-5, FUS/TLS, G-CSFR, G9a, GABA T-1, GABA
B R1, GABAA Rγ3, GABAA Rγ4, GABAB R1β, GABARAPL1, GAD-65, GAD-67, GADL1, GAGE10, GAGE3, GalNAc-TL2, GCFC1, GCN5, GDE2, GDF-11, GDF5OS, Gemin2, Gemin4, Gigaxonin, GLDC, Gle1, gliacolin, Gliomedin, GLIPR1, GLIS1, GLT8D1, GLTSCR1, Glutamate-Pyruvate Transaminase, glycogen synthase 1, GlyR α4, Gm14137, GnT-II, GON4L, GPR101, Gpr165, GPR177, GPR85, GRHL2, Grid2ip, GSC2, GSH-2, GSKIP, GTF2IRD2B, H1FOO, H1T2, H2-Kd, H2-M10.1, H2-M10.3, H2-M11, H2-M9, H2-Q6, H2-T24, H2AL1, H2AL2, H2al2y, H2BFA, H3.Y histone 1, HABP2, Hairless, Hairy Cell Leukemia Marker, HAT1, Hat2, HATH-6, HB9, Hbb-b2, Hbb-y, HBO1, hCAP-D3, HCP1, HDA1, HDA4, HDA5, HDA6, HDA7, Hdf1, HEB, HEL308, Helios, HEMK1, Hep B preS1, HERP, HES3, HES4, HES5, HES7, HESX1, HHAT, HHIPL2, HHV-4 EBNA-2, HHV-4 EBNA-3A, HHV-4 EBNA-LP, HHV-4 ZEBRA, HHV-6, HHV-8, Hils1, HIP2, Histone cluster 1 H1B, Histone cluster 1 H2A, Histone cluster 1 H2AA, Histone cluster 1 H2AA2, Histone cluster 1 H2AB, Histone cluster 1 H2AG, Histone cluster 1 H2AI, Histone cluster 1 H2AJ, Histone cluster 1 H2AO, Histone cluster 1 H2B, Histone cluster 1 H2BA, Histone cluster 1 H2BC, Histone cluster 1 H2BD, Histone cluster 1 H2BG, Histone cluster 1 H2BL, Histone cluster 1 H2BM, Histone cluster 1 H2BP, Histone cluster 1 H3F, Histone cluster 1 H3H, Histone cluster 1 H3I, Histone cluster 1 H4, Histone cluster 1 H4G, Histone cluster 1 H4H, Histone cluster 1 H4I, Histone cluster 1 H4M, Histone cluster 2 H2AA1, Histone cluster 2 H2BE, Histone cluster 2 H2BF, Histone cluster 2 H3A, Histone cluster 2 H3B, Histone cluster 2 H3C1, Histone cluster 2 H3C2, Histone cluster 2 H4, Histone cluster 2 H4A, Histone cluster 3 H2ba, Histone cluster 3 H2BB, Histone cluster 4 H4, Histone H1B, Histone H1X, Histone H2A, Histone H2A.F, Histone H2A.V, Histone H2A.Z, Histone H3, Histone H3.3A, Histone Z, HIV-1 Gag, HIV-1 p17, HIV-1 p55, HLA-A2, HLA-E, HLA-F`, HLF, HMG I, HMG-14, HMG-4, HMG20A, HMP-2, hnRNP A/B, hnRNP A2/B1, hnRNP C, hnRNP H′/hnRNP H2, Homer-1, HOS, HoxA3, HoxA9, HoxB1, HoxB4, HoxB6, HoxD1, HoxD10, HoxD4, HoxD9, HP1α, HP1β, HP1γ, HRG4, HRT1_Hrt1, hSNF2H, HSPC171, HSV-1 ICP0, HSV-2 ICP10, HTLV-1 gp46, HTLV-1 p19, HTR3E, HuD, Huntingtin, I830077J02Rik, IDAS, IFN-δ, IFT122, IGSF10, IKAP, Ikaros 5, IL-13Rα2, ILEI, ILKAP, IMP-3, IMPA1, IMPA2, IMPA3, Ini1, INO80D, INSM1, INTS4, IP3KA, IQSEC1, IRX6, Isw1p, Iws1, JAB1, Jagged1, Jagged2, JIP-4, JMJD5, Kaiso, KALIG-1, Kar2, karyopherin α2, karyopherin α3, karyopherin 13, KAT I, KAT III, KCNH8, KcsA, KCTD11, KCTD17, KCTD19, KCTD3, Khdc1a, Khdc1c, KIAA0146, KIAA1024L_A730017C20Rik, KIAA1688, KIAA1704, KIAA2022, KIAA2026_9930021J03Rik, KIDINS220, KIR3DL2, KIR3DL3, kismet-L, kleisin β, KLF7, Klra33, KRBOX1, Kremen-1, Kremen-2, Krox-26, KRTAP12-3, KRTAP2-2, KSHV ORF 45, Kua-Uev, KV2.1, L1TD1, L3MBTL2, LAGE-2A, Lamin A/C, Lamin B2, LAMP-1, LAMP-2, LANPL, LAP2, LBH, LBX1, LBXCOR1, LCOR, LDOC1L, LEF-1, Lefty, LEKR1_Lekr1, LEUTX, LGI2, LGR6, LHFP, LHX1, LHX3, LHX8, LIMCH1, LIN-15B, LIN-28B, LIN-44, LIN-54, LINE-1, Liprin α4, LIS1, LIX1, LMX1B, LOC100039210, LOC100039269, LOC100039596, LOC100039674, LOC100039794, LOC100039815, LOC100039830, LOC100039899, LOC100040201, LOC100040299, LOC100040500, LOC100040682, LOC100040839, LOC100040923, LOC100041102, LOC100041177, LOC100041224, LOC100041241, LOC100041253, LOC100041281, LOC100041317, LOC100041351, LOC100041407, LOC128977, LOC345643, LOC390245, LOC439950, LOC439994, LOC440233, LOC440563, LOC440900, LOC642366, LOC643072, LOC643623, LOC644192, LOC644248, LOC644838, LOC645359, LOC646522, LOC646817, LOC730159, LRRC1, LRRC52, LRRFIP1, LRTM1, Lunatic Fringe, LYPD3, LZIC, M-cadherin, M33, Mael, MAGE, MAGE-A4, MAGE-A5, MAGE-A6, MAGE-E1, MAGE-F1, MAGE-G2, MAN1B1, Manic Fringe, MAP-1A, MAP-2A, MAP-2B, MAP-2C, MATH-1, matrin-3, MAU-2, MAVS, MBD3, MBD5, MBTD1, Mcl-1, MDGA1, MDGA2, MeCP2, Med2p, Med4, MEIOB, melanotransferrin, MESP1, Mfn1, MFSD9, Mg29, MGC1203, mGluR-7, MLLT6, MOF, MOP-3, MORF4L1, MPc3_CBX8, MPHOSPH8, MRFAP1, MRG1, MRG15, MRGBP, MRGX2, mSin3A, MSL-1, MTAP, MTF-2, MUSTN1, Mxi 1, Myo-inositol monophosphatase 1, MyoD, Myosin Ih, MYT1, MYT1L, N28178, Na+ CP type Iβ, NaDC-3, NAGS, NALP5, Nanos2, NAP1L3, NAP1L4, NAPRT, NAT-9, NBPF1, NBPF15, NBPF16, NBPF24, NBPF8, Ncapd3, NEIL1, NEPH2, nephrocystin-5, NESP55, Nestin, Neurensin-1, neurexin II, neurexin III, Neurexophilin-2, Neuritin, NEURL, Neuro D, Neurobeachin, Neurofascin, Neurofibromin, Neurogenin 1, neuroligin 1, neuroligin 2, neuroligin X/Y, Neuromedin N, Neurotrimin, Neurotrypsin, NF-L, NF-YB, NFAT5, NFATc2IP, NKG2-A, Nkx-6.2, NLE1, nm23-H1, NOMO2, NOMO3, Nop58, Norpeg, NPAS3, NPAS4, NPW, NRSF, NSBP1, NSD2, NUDE1, NUDT16, NUT, Oct-3/4, Oct-3A, Oct-4, OCTL2, Olfr702, Olfr710, Olfr711, Olfr713, Olfr826, OLIG1, OLIG3, OPA1, OR10K1, OR13G1, OR1M1, OR2L3, OR2T10, OR4F16, OR52P1, OR5H15, OR6Y1, Orexin R-2, Orf Virus, OTTMUSG00000002180, OTTMUSG00000004968, OTTMUSG00000005148, OTTMUSG00000005215, OTTMUSG00000010552, OTTMUSG00000010657, OTTMUSG00000010747, OTTMUSG00000011097, OTTMUSG00000016607, OTTMUSG00000016611, OTTMUSG00000016698, OTTMUSG00000016703, OTTMUSG00000016780, OTTMUSG00000016781, OTTMUSG00000016783, OTTMUSG00000016784, OTTMUSG00000016785, OTTMUSG00000016788, OTTMUSG00000016789, OTTMUSG00000016790, OTTMUSG00000018481, OTX1, P2Y13, p32, p57, P5CR2, PA28α, PABPN1, Paralemmin, Pax-3, Pax-5, Pbx 4, PC2, PCAF, PCDH20_Pcdh20, PCDHA11, PCDHA12, PCDHA2, PCDHAC, PCDHAC1_Pcdhac1, PCDHAC2_Pcdhac2, PCDHB1, PCDHB2, Pcdhb22, PCDHBPcdhb22, PCDHGA1, PCDHGA2, PCDHGA3, PCDHGA4, PCDHGA5, PCDHGA7_Pcdhga7, PCDHGA8, PCDHGA9, PCDHGB1, PCDHGB2, Pcdhgb4, PCDHGB5, PCDHGB6, PCDHGC4, Pcf11, PCM1, PCNXL2, PCP-4, PCP4L1, PDIK1L, Peg12, PEPT2, Per3, Periostin, Peripherin, PGAM4, PGPEP1L, PHAX, PHC1, PHC2, PHF13, PHF20, PHF21B, PHF7, PHF8, PHKB, PLAA, plasmolipin, PNKD, PNMAL1, POLR3A, Polycomb, POTEJ, Ppp4r1l, PRAME like-3, PRAME like-4, PRAME like-6, PRAME like-7, PRAMEF1, PRAMEF10, PRAMEF12, PRAMEF16, PRAMEF17, PRAMEF2, PRAMEF20, PRAMEF23, PRAMEF4, PRDM10, PRDM8, Presenilin 1, Prickle2, Prickle4, PRIP, Prl2c1, PRR19, PRR23A, PRRT3, PSAP, PSD-95, PTF1, PTH2 Receptor, Purγ, Pygopus 1, Pygopus 2, pyridoxal phosphatase, R-cadherin, R-Spondin, R-Spondin2, R-Spondin3, R3HCC1, Rab GAP1L, Rab L4, Rab L5, Radical Fringe, RAG-2, RAI2, RALGAPB, RASSF1, RBA-1, RbAp46, RbAp48, RBFOX3/NeuN, RBP-Jκ, RDS, Reelin, RFLAT-1, RFRP-2, RGPD8, RhC, Rho T2, RHOBTB3, Rhox1, Rhox10, Rhox11, Rhox12, Rhox13, Rhox2b, Rhox2c, Rhox2d, Rhox2e, Rhox3a, Rhox3f, Rhox3g, Rhox3h, Rhox4b, Rhox4c, Rhox4d, Rhox4e, Rhox4f, Rhox4g, RNF123, RNF20, RP11-430L17.1, RP11-592B15.4, Rpb3, Rpb4, Rsl1, RSRC1, RTP1, RUSC1, S-100 β chain, Sall3, SAP 25_Sap25, SAP 30L, SAPAP3, SBDS, SBF2, SBP Tag, SBZF3, SCAND1, SCML2, SEB, SEBOX, SEC22A, Sec24, Septin 6, Serpinb6d, Set1, SEZ6, SFMBT1, SFRS8, Shank 3, SHOX2B, Sialyltransferase 7A, Sidekick-1, Siglec-9, Six6, SKT, Slfn5, SLITRK2, SLITRK6, Slxl1, Sm B/B′, Smad7, Smarcad1, SMARCD2, SMARCD3, SMC3, SMCHD1, SmcX, SNF2L, SNURF, SOCS-5, SorCS3, Sox-1, Sox-10, Sox-15, Sox-2, Sox-21, Sox-22, Sox-9, SP140, SP140L, Sp4, Sp6, SPATA21, SPEN, SPF30, SPINK2, SPT16, SR-1F, SR-2C, SRE-ZBP, SRGAP2P1, SRp75, SRPX2, SSEA-1, SSEA-3, SSEA-4, SSRP1, SSX1, SSX2, STAF42, STOX2, STPG1, Stra8, SUCLA2, Sum1, SURF-2, Survivin, SUV420H2, SUZ12, Swe1, SWI/SNF-B, Swi3, Sycp2l, syncollin, Syntaphilin, Syntaxin 16, Syntaxin 1A, SYP, TADA2L, TADA3L, TAF II p140, TAF5L, TANC2, TARDBP, Tau, TBRG4, TBX2, TBX5, TBX6, TCEA1, TCF-1, TCF-19, TCF-4, TCFL5, TCTA, Tctex3, Teneurin-1, TERC, TESSP5, TEX19, TFIIA-β, THAP10, THAP7, THAP8, TIG1, TIGD2, TIP60, TLE1, TLE5, TMC2, TMEM145, TMEM147, TMEM200C, TMEM97, TMPPE, TMPRSS11E, TNP2, TRA-1-81, TRAAK, TRIM2, TRIM27, TRIM32, Trimethyl Histone H3, Trk B, TRPS1, Trypsin, TSR3, TTC14, TTLL7, TTMP_BC030499, Twinkle, Twist, u4/u6 snRNP, Ub, UBE2F, UBN-2, UBXD3, UCH-L1, UGT2B7, UHRF1, ULBP5, UNC-2, UNCX, URB2, USH1G, USP20, USP28, UTF1, utrophin, VAP-B/C, Varicella Zoster Virus gpIII, Varicella Zoster Virus IE62, VCPIP1, VGLUT3, VILL, Vmn1r1, Vmn1r12, Vmn1r175, Vmn1r31, VP16, VPS72, VSV-G, VWC2L, VZV IE62, VZV Major Capsid Protein, WDR11, WDR5B, WDR65, WDSUB1, Wingless, WIPI-3, Wnt-1, Wnt-10a, Wnt-10b, Wnt-14, Wnt-15, Wnt-16, Wnt-2, Wnt-3a, Wnt-5a, Wnt-5b, Wnt-7a, Wnt-7b, Wnt-8a, WSTF, XAGE-1B, XBP-1, YAF2, YDJC, YPEL1, ZBBX_FLJ23049, ZBTB24, ZBTB25, ZBTB4, ZBTB8, ZCCHC18, ZCCHC2, ZCCHC3, ZDHHC18, ZDHHC19, ZFHX2, ZFP106, ZFP161, ZFP41, ZFP773, Zfp874, ZFY, ZGPAT, Zic1, Zic5, ZKSCAN2, ZMAT2, ZNF143, ZNF157, ZNF16, ZNF174, ZNF197, ZNF198, ZNF228, ZNF229, ZNF250, ZNF251, ZNF258, ZNF281, ZNF295, ZNF300, ZNF312, ZNF318, ZNF322A, ZNF337, ZNF33A, ZNF33B, ZNF34, ZNF341, ZNF343, ZNF345, ZNF382, ZNF445, ZNF462, ZNF469, ZNF498, ZNF502, ZNF508, ZNF510, ZNF519, ZNF524, ZNF532, ZNF536, ZNF569, ZNF572, ZNF585B, ZNF597, ZNF619, ZNF621, ZNF623, ZNF625, ZNF643, ZNF644, ZNF648, ZNF662, ZNF669, ZNF671, ZNF696, ZNF7, ZNF707, ZNF709, ZNF729, ZNF746, ZNF766, ZNF808, ZNF81, ZNF814, ZNF828, ZNF831, ZNF852, ZNHIT1, ZNHIT6, ZSCAN18, ZSWIM3, and ZSWIM6.