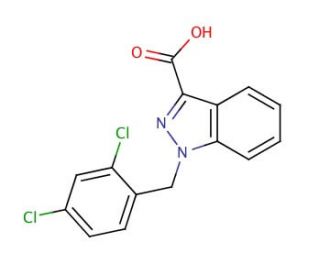
Molecular structure of Lonidamine, CAS Number: 50264-69-2
Lonidamine (CAS 50264-69-2)
See product citations (7)
Alternate Names:
1-(2,4-Dichlorobenzyl)-1H-indazole-3-carboxylic acid
Application:
Lonidamine is it elicits cell apoptosis and blocks CFTR Cl- channel
CAS Number:
50264-69-2
Purity:
≥99%
Molecular Weight:
321.16
Molecular Formula:
C15H10Cl2N2O2
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Lonidamine is an indazole compound that has been reported to inhibit angiogenic-related endothelial cell functions. This compound has been found to trigger apoptosis in endothelial cells and cause a decrease in proliferation, migration, invasion, and morphogenesis. The apoptotic effects of Lonidamine have been demonstrated to be due to Bax, Bid, caspase-3, and caspase-9 activation, as well as cytochrome c release. Futher studies show that Lonidamine prevents cellular energy metabolism via HXK (hexokinase) inhibition. This compound also blocks CFTR Cl- channels in vitro.
Lonidamine (CAS 50264-69-2) References
- Recent studies on lonidamine, the lead compound of the antispermatogenic indazol-carboxylic acids. | Gatto, MT., et al. 2002. Contraception. 65: 277-8. PMID: 12020777
- Mechanism of lonidamine inhibition of the CFTR chloride channel. | Gong, X., et al. 2002. Br J Pharmacol. 137: 928-36. PMID: 12411425
- Lonidamine: efficacy and safety in clinical trials for the treatment of solid tumors. | Di Cosimo, S., et al. 2003. Drugs Today (Barc). 39: 157-74. PMID: 12730701
- Lonidamine causes inhibition of angiogenesis-related endothelial cell functions. | Del Bufalo, D., et al. 2004. Neoplasia. 6: 513-22. PMID: 15548359
- Curcumin stimulates reactive oxygen species production and potentiates apoptosis induction by the antitumor drugs arsenic trioxide and lonidamine in human myeloid leukemia cell lines. | Sánchez, Y., et al. 2010. J Pharmacol Exp Ther. 335: 114-23. PMID: 20605902
- Reviving Lonidamine and 6-Diazo-5-oxo-L-norleucine to Be Used in Combination for Metabolic Cancer Therapy. | Cervantes-Madrid, D., et al. 2015. Biomed Res Int. 2015: 690492. PMID: 26425550
- Re-programming tumour cell metabolism to treat cancer: no lone target for lonidamine. | Bhutia, YD., et al. 2016. Biochem J. 473: 1503-6. PMID: 27234586
- Targeting lonidamine to mitochondria mitigates lung tumorigenesis and brain metastasis. | Cheng, G., et al. 2019. Nat Commun. 10: 2205. PMID: 31101821
- Targeting glucose metabolism to suppress cancer progression: prospective of anti-glycolytic cancer therapy. | Abdel-Wahab, AF., et al. 2019. Pharmacol Res. 150: 104511. PMID: 31678210
- Lonidamine potentiates the oncolytic efficiency of M1 virus independent of hexokinase 2 but via inhibition of antiviral immunity. | Cai, J., et al. 2020. Cancer Cell Int. 20: 532. PMID: 33292203
- Multiple targeted doxorubicin-lonidamine liposomes modified with p-hydroxybenzoic acid and triphenylphosphonium to synergistically treat glioma. | Lu, J., et al. 2022. Eur J Med Chem. 230: 114093. PMID: 35007860
- Lonidamine and domperidone inhibit expansion of transformed cell areas by modulating motility of surrounding nontransformed cells. | Aoyama, M., et al. 2022. J Biol Chem. 298: 102635. PMID: 36273581
- Effects of Lonidamine on murine and human tumor cells in vitro. A morphological and biochemical study. | De Martino, C., et al. 1984. Oncology. 41 Suppl 1: 15-29. PMID: 6717891
Inhibitor of:
3 BP-1, ACSVL5, C1orf112, CARKL, CRIF1, D10Wsu102e, DNTTIP2, EG433182, ENO4, ETFDH, FBP, G3PP, GLYATL3, GLYCTK, Hex, Hexokinase, hnRNP E3, HXK, HXK I, HXK II, HXK III, IDHC, IER2, Inhibitors, LDHC, MCT1, MCT11, MCT12, MCT2, MCT4, ME1, NDUFAB1, NDUFB3, PDPR, PFK-1, PFK-2 tes, PFKP, PGAM1, PHD, SCOCO, Sperm-associated antigen (SPAG), TKTL1 (transketolase), TKTL2, and XYLB.Activator of:
caspase-3, caspase-9, and VDAC2.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lonidamine, 5 mg | sc-203115 | 5 mg | $105.00 | |||
Lonidamine, 25 mg | sc-203115A | 25 mg | $364.00 |
