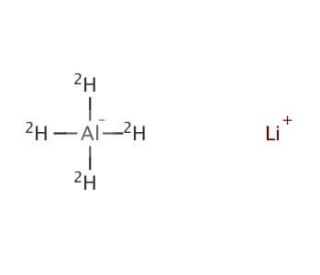

Molecular structure of Lithium Aluminum Deuteride
Lithium Aluminum Deuteride (CAS 14128-54-2)
Alternate Names:
Tetrahydro-d4-Aluminate(1-) Lithium; Aluminum Lithium Hydride (LiAlD4); Aluminum Lithium Hydride (AlLiD4)
Application:
Lithium Aluminum Deuteride is used in various analytical organic chemistry studies
CAS Number:
14128-54-2
Molecular Weight:
41.98
Molecular Formula:
AlD4Li
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
Available in US only.
Available in US only.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Lithium aluminium deuteride possesses multiple applications in scientific research, encompassing isotope labeling, deuterium exchange reactions, and deuterium isotope effects. Additionally, it serves as a deuterium source for synthesizing various compounds, including organometallic compounds. This compound exhibits remarkable stability and low reactivity, making it an ideal deuterium source for numerous scientific research endeavors. Through reactions, the deuterium atoms within the compound undergo exchange with other atoms, leading to the creation of novel compounds.
Lithium Aluminum Deuteride (CAS 14128-54-2) References
- Synthesis of 4-d1-testosterone and 4-d1-androstenedione. | Nambara, T., et al. 1976. Chem Pharm Bull (Tokyo). 24: 2486-93. PMID: 1017088
- Biotransformation of cannabidiol in mice. Identification of new acid metabolites. | Martin, BR., et al. 1977. Drug Metab Dispos. 5: 259-67. PMID: 17524
- Synthesis of spirocyclic indolines by interruption of the Bischler-Napieralski reaction. | Medley, JW. and Movassaghi, M. 2013. Org Lett. 15: 3614-7. PMID: 23829389
- Synthesis of alpha-deuterium-labelled cyclohexylamine and its deamination by rabbit liver microsomes. | Kurebayashi, H., et al. 1989. Chem Pharm Bull (Tokyo). 37: 1097-9. PMID: 2766411
- Analysis by the reductive-cleavage method of linkage positions in a polysaccharide containing 4-linked D-glucopyranosyluronic residues. | Vodonik, SA. and Gray, GR. 1988. Carbohydr Res. 175: 93-102. PMID: 3288343
- Detection of the in vivo conversion of 2-pyrrolidinone to gamma-aminobutyric acid in mouse brain. | Callery, PS., et al. 1979. Biomed Mass Spectrom. 6: 23-6. PMID: 427258
- Mass spectra of various deuterium-labelled forms of bis-O-trimethylsilyl-N-acetylsphinganine. | Krisnangkura, K. and Sweeley, CC. 1974. Chem Phys Lipids. 13: 415-28. PMID: 4452220
- The structure of mycoheptin, a polyene macrolide antifungal antibiotic. | Borowski, E., et al. 1978. J Antibiot (Tokyo). 31: 117-23. PMID: 632222
- Deuterium isotope effects on the enzymatic oxidative deamination of trace amines. | Yu, PH., et al. 1981. Biochem Pharmacol. 30: 3089-94. PMID: 7337725
- Chloroperoxidase-catalyzed benzylic hydroxylation. | Miller, VP., et al. 1995. Arch Biochem Biophys. 319: 333-40. PMID: 7786013
- The Mechanism of Lithium Aluminum Hydride Double-Bond Reductions in the 7-Substituted Norbornadienes and syn-7-Substituted Norbornenes1 | Boris Franzus and Eugene I. Snyder. 1965. J. Am. Chem. Soc. 87, 15: 3423–3429.
- Organic ions in the gas phase. XIX. Hydrogen randomization in gaseous C7H7+ ions | Seymour Meyerson, Harold Hart, and L. C. Leitch. 1968. J. Am. Chem. Soc. 90, 13: 3419–3423.
- A convenient synthetic sequence for the deuterium labeling of olefins in the allylic position | M. Bellarmine Grdina, Michael Orfanopoulos, and L. M. Stephenson. 1979. J. Org. Chem. 44, 16: 2936–2938.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lithium Aluminum Deuteride, 1 g | sc-488305 | 1 g | $550.00 | |||
| US: Only available in the US | ||||||
Lithium Aluminum Deuteride, 5 g | sc-488305A | 5 g | $1650.00 | |||
| US: Only available in the US | ||||||
