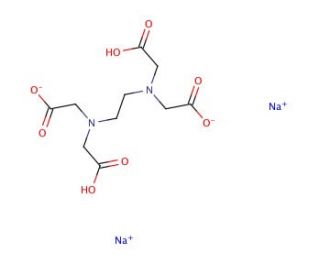
Ethylenediaminetetraacetic acid disodium salt, 0.050M Standardized solution (CAS 139-33-3)
QUICK LINKS
Ethylenediaminetetraacetic acid (EDTA) disodium salt in a standardized 0.050M solution is a crucial reagent extensively utilized in various research applications due to its chelating properties and versatile nature. As a powerful chelating agent, EDTA forms stable complexes with metal ions by binding to them through its multiple amine and carboxylate groups. This mechanism enables EDTA to effectively sequester metal ions, thereby preventing them from participating in undesirable chemical reactions or catalyzing oxidative processes. In research, EDTA finds widespread use in analytical chemistry, particularly in complexometric titrations for the determination of metal ions in solution. Its ability to form stable complexes with metal ions makes it invaluable in the determination of metal concentrations in environmental samples, food products, and biological fluids. Moreover, EDTA is employed in molecular biology and biochemistry for its role in nucleic acid isolation and purification. By chelating divalent metal ions, EDTA helps to inhibit nucleases, which could otherwise degrade DNA and RNA during extraction and storage processes. Furthermore, EDTA is utilized in cell culture applications to chelate metal ions in cell culture media, thus preventing metal-mediated cytotoxicity and maintaining cell viability. Overall, the standardized EDTA solution serves as a fundamental tool in numerous research endeavors across various scientific disciplines, facilitating precise analytical measurements and ensuring the integrity of experimental outcomes.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethylenediaminetetraacetic acid disodium salt, 0.050M Standardized solution, 1 L | sc-397341 | 1 L | $45.00 | |||
Ethylenediaminetetraacetic acid disodium salt, 0.050M Standardized solution, 4 L | sc-397341A | 4 L | $85.00 |
