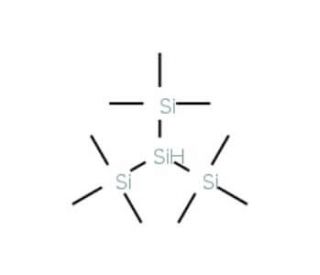
Tris(trimethylsilyl)silane (CAS 1873-77-4)
QUICK LINKS
Tris(trimethylsilyl)silane (TMS) is a versatile organosilicon compound extensively utilized in chemical synthesis, scientific research, and laboratory experiments. This volatile liquid offers the ability to modify the physical and chemical properties of various substances such as polymers, plastics, and organometallic compounds. It has found broad applications in fields including organic chemistry, biochemistry, and materials science. In scientific research, Tris(trimethylsilyl)silane has proven valuable as a reagent for synthesizing organometallic compounds, polymers, and pharmaceuticals. It also functions as a catalyst in polymer synthesis and serves as a protective agent during the formation of organometallic compounds. Moreover, its applications extend to the study of protein structure, function, enzyme kinetics, and enzymology. Tris(trimethylsilyl)silane acts as a Lewis acid, exhibiting the ability to form covalent bonds with electron-rich molecules such as alcohols and amines. This Lewis acidity enables it to catalyze the synthesis of diverse organometallic compounds and polymers. Furthermore, it can act as a protective agent, preventing undesired side reactions during the synthesis of organometallic compounds.
Tris(trimethylsilyl)silane (CAS 1873-77-4) References
- Radical-mediated silyl- and germyldesulfonylation of vinyl and (alpha-fluoro)vinyl sulfones: application of tris(trimethylsilyl)silanes and tris(trimethylsilyl)germanes in Pd-catalyzed couplings. | Wnuk, SF., et al. 2004. Org Lett. 6: 2047-9. PMID: 15176815
- Tris(trimethylsilyl)silane: an unprecedented enhancement in the diastereoselectivity of radical cyclisations to give 2,4-disubstituted piperidines. | Gandon, LA., et al. 2004. Org Biomol Chem. 2: 2270-1. PMID: 15305204
- Regioselective hydrosilylations of propiolate esters with tris(trimethylsilyl)silane. | Liu, Y., et al. 2005. J Org Chem. 70: 556-61. PMID: 15651801
- Synthesis of 2,4-disubstituted piperidines via radical cyclization: unexpected enhancement in diastereoselectivity with tris(trimethylsilyl)silane. | Gandon, LA., et al. 2006. J Org Chem. 71: 5198-207. PMID: 16808507
- Tris(trimethylsilyl)silane (TTMSS)-derived radical reactivity toward alkenes: a combined quantum mechanical and laser flash photolysis study. | Lalevée, J., et al. 2007. J Org Chem. 72: 6434-9. PMID: 17658850
- Safe, facile radical-based reduction and hydrosilylation reactions in a microreactor using tris(trimethylsilyl)silane. | Odedra, A., et al. 2008. Chem Commun (Camb). 3025-7. PMID: 18688336
- Hypersilylated cyclodiphosphadiazanes and cyclodiphosphadiazenium salts. | Kuzora, R., et al. 2009. Dalton Trans. 9304-11. PMID: 20449210
- Recent applications of the (TMS)3SiH radical-based reagent. | Chatgilialoglu, C. and Lalevée, J. 2012. Molecules. 17: 527-55. PMID: 22269866
- Tris(trimethylsilyl)silane and visible-light irradiation: a new metal- and additive-free photochemical process for the synthesis of indoles and oxindoles. | da Silva, GP., et al. 2015. Chem Commun (Camb). 51: 15110-3. PMID: 26324353
- Synthesis of Ultrathin Copper Nanowires Using Tris(trimethylsilyl)silane for High-Performance and Low-Haze Transparent Conductors. | Cui, F., et al. 2015. Nano Lett. 15: 7610-5. PMID: 26496181
- Tris(trimethylsilyl)silane as a co-initiator for dental adhesive: Photo-polymerization kinetics and dynamic mechanical property. | Song, L., et al. 2016. Dent Mater. 32: 102-13. PMID: 26616688
- DFT Mechanistic Study of IrIII/NiII-Metallaphotoredox-Catalyzed Difluoromethylation of Aryl Bromides. | Xu, J., et al. 2021. Inorg Chem. 60: 8682-8691. PMID: 34110137
- Physicochemical properties of experimental resin-based composites using tris(trimethylsilyl)silane associated with diphenyl iodonium hexafluorophosphate (DPIHP) salt. | Nobre, CFA., et al. 2022. J Mech Behav Biomed Mater. 130: 105169. PMID: 35305411
- Sustainable Protocol for the Synthesis of 2',3'-Dideoxynucleoside and 2',3'-Didehydro-2',3'-dideoxynucleoside Derivatives. | Martín-Nieves, V., et al. 2022. Molecules. 27: PMID: 35807233
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Tris(trimethylsilyl)silane, 5 g | sc-296666 | 5 g | $133.00 | |||
Tris(trimethylsilyl)silane, 25 g | sc-296666A | 25 g | $329.00 |
