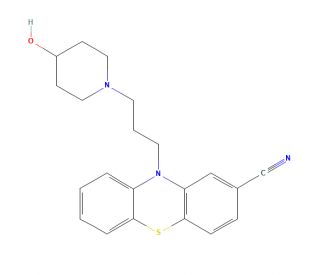
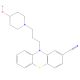
Pericyazine (CAS 2622-26-6)
QUICK LINKS
Pericyazine is a compound studied in various research applications, particularly within the scope of its interactions with neurotransmitter systems. In neurochemical studies, Pericyazine is of interest for its effects on dopamine receptors, where it is used to understand the modulation of dopaminergic signaling pathways. This compound is also employed in the examination of receptor binding profiles, helping to elucidate the mechanisms by which it influences neurotransmitter activity. Additionally, Pericyazine serves as a tool in the study of synaptic transmission and is used to observe changes in neurotransmitter release and reuptake under different experimental conditions. Researchers may also investigate the impact of Pericyazine on cellular models to gain insights into its molecular mechanisms of action.
Pericyazine (CAS 2622-26-6) References
- Development of predictive retention-activity relationship models of antipsychotic drugs by micellar liquid chromatography. | Martín-Biosca, Y., et al. 1999. Biomed Chromatogr. 13: 478-92. PMID: 10534760
- An LD50 model for predicting psychotropic drug toxicity using biopartitioning micellar chromatography. | Quiñones-Torrelo, C., et al. 2001. Biomed Chromatogr. 15: 31-40. PMID: 11180298
- A simple high-performance liquid chromatographic method for detection of hydroxyzine in human plasma after overdose. | Péhourcq, F. 2004. J Pharmacol Toxicol Methods. 50: 41-4. PMID: 15233966
- Cloud point extraction and preconcentration for the gas chromatography of phenothiazine tranquilizers in spiked human serum. | Ohashi, A., et al. 2004. Anal Sci. 20: 1353-7. PMID: 15478348
- Toxicological screen on gastric aspirates: the Singapore experience. | Chao, TC., et al. 1992. Forensic Sci Int. 54: 141-51. PMID: 1639280
- Identification and quantification of 30 antipsychotics in blood using LC-MS/MS. | Saar, E., et al. 2010. J Mass Spectrom. 45: 915-25. PMID: 20648676
- Inhibition of hepatic drug metabolism by phenothiazine tranquilizers: quantitative structure-activity relationships and selective inhibition of cytochrome P-450 isoform-specific activities. | Murray, M. 1989. Chem Res Toxicol. 2: 240-6. PMID: 2577523
- Expression and oxidative modifications of plasma proteins in autism spectrum disorders: Interplay between inflammatory response and lipid peroxidation. | Cortelazzo, A., et al. 2016. Proteomics Clin Appl. 10: 1103-1112. PMID: 27246309
- Doping control analysis of 121 prohibited substances in equine hair by liquid chromatography-tandem mass spectrometry. | Wong, JKY., et al. 2018. J Pharm Biomed Anal. 158: 189-203. PMID: 29885604
- Mapping human microbiome drug metabolism by gut bacteria and their genes. | Zimmermann, M., et al. 2019. Nature. 570: 462-467. PMID: 31158845
- Effect of neuroleptics on indoleamine-N-methyltransferase activity and brain metabolism of bufotenin. | Gomes, UC., et al. 1981. Biochem Pharmacol. 30: 571-5. PMID: 6115652
- Relationship between the ability of some neuroleptics to enhance striatal [3H]dopamine release and their lipophilicity. | Goosey, MW. and Doggett, NS. 1983. Biochem Pharmacol. 32: 2411-6. PMID: 6137221
- Acetylcholinesterase activities in cerebrospinal fluid of patients with senile dementia of Alzheimer type. | Soininen, H., et al. 1981. Acta Neurol Scand. 64: 217-24. PMID: 7315192
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Pericyazine, 25 mg | sc-478905 | 25 mg | $360.00 | |||
Pericyazine, 250 mg | sc-478905A | 250 mg | $2503.00 |
