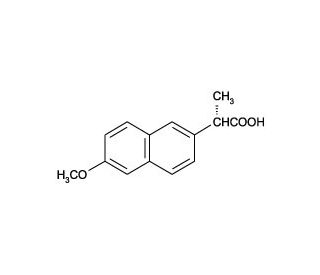
Molecular structure of Naproxen, CAS Number: 22204-53-1
Naproxen (CAS 22204-53-1)
Alternate Names:
(S)-(+)-2-(6-Methoxy-2-naphthyl)propionic Acid;(S)-(+)-6-Methoxy-α-methyl-2-naphthaleneacetic Acid
Application:
Naproxen is a Cox-1 and Cox-2 inhibitor
CAS Number:
22204-53-1
Purity:
≥98%
Molecular Weight:
230.26
Molecular Formula:
C14H14O3
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Naproxen is widely used in research focused on its effects on enzyme activity, specifically its interaction with cyclooxygenase enzymes. This interaction is critical for understanding the mechanisms of enzyme inhibition and regulation. In biochemical studies, Naproxen is often utilized to investigate the pathways involved in inflammatory responses at the molecular level. Researchers also use Naproxen to study its influence on the production of prostaglandins and other chemical messengers within various tissues. Additionally, it serves as a tool in experiments aimed at understanding the modulation of biochemical pathways involved in pain and inflammation responses.
Naproxen (CAS 22204-53-1) References
- Molecular basis for cyclooxygenase inhibition by the non-steroidal anti-inflammatory drug naproxen. | Duggan, KC., et al. 2010. J Biol Chem. 285: 34950-9. PMID: 20810665
- Interaction of copper(II) with the non-steroidal anti-inflammatory drugs naproxen and diclofenac: synthesis, structure, DNA- and albumin-binding. | Dimiza, F., et al. 2011. J Inorg Biochem. 105: 476-89. PMID: 20926136
- Nonsteroidal anti-inflammatory drug naproxen destabilizes Aβ amyloid fibrils: a molecular dynamics investigation. | Takeda, T., et al. 2010. J Phys Chem B. 114: 15394-402. PMID: 20979356
- Naproxen in the horse: pharmacokinetics and side effects in the elderly. | Della Rocca, G., et al. 2014. Res Vet Sci. 96: 147-52. PMID: 24388762
- Effect of R and S enantiomers of naproxen on aggregation and thromboxane production in human platelets. | Kean, WF., et al. 1989. J Pharm Sci. 78: 324-7. PMID: 2724096
- Synthesis of naproxen-imprinted polymer using Pickering emulsion polymerization. | Kujawska, M., et al. 2018. J Mol Recognit. 31: PMID: 28322471
- Chiral recognition of naproxen enantiomers based on fluorescence quenching of bovine serum albumin-stabilized gold nanoclusters. | Jafari, M., et al. 2017. Spectrochim Acta A Mol Biomol Spectrosc. 185: 77-84. PMID: 28549294
- Naproxen and Its Phototransformation Products: Persistence and Ecotoxicity to Toad Tadpoles (Anaxyrus terrestris), Individually and in Mixtures. | Cory, WC., et al. 2019. Environ Toxicol Chem. 38: 2008-2019. PMID: 31403235
- Naproxen in the environment: its occurrence, toxicity to nontarget organisms and biodegradation. | Wojcieszyńska, D. and Guzik, U. 2020. Appl Microbiol Biotechnol. 104: 1849-1857. PMID: 31925484
- Anticancer and Antimicrobial Activities of Naproxen and Naproxen Derivatives. | Han, Mİ. and Küçükgüzel, ŞG. 2020. Mini Rev Med Chem. 20: 1300-1310. PMID: 32368976
- Recent Advances in the Synthesis of Ibuprofen and Naproxen. | Ha, MW. and Paek, SM. 2021. Molecules. 26: PMID: 34443379
- Preparation, Characterization and Study of the Dissociation of Naproxen from Its Chitosan Salt. | Medeiros, RS., et al. 2022. Molecules. 27: PMID: 36144537
- Effect of naproxen on the model lipid membrane formed on the water-chitosan subphase. | Przykaza, K., et al. 2023. Biochim Biophys Acta Biomembr. 1865: 184099. PMID: 36493856
Inhibitor of:
9530003J23Rik, AADAC, AKR1C12, AKR1C13, AKR1C14, AKR1C18, AKR1C19, AKR1C20, AKR1C6, Aldo-keto reductase (AKR) family, AOX1, APOBEC3H, ATP5H, BSA, BTR1, C6, Complexin-1, Cox-2, CTL2, CysLT1 Receptor, cytohesin-4, D11Wsu47e, DBX1, DD1, ENDOGL1, Enzyme, ESAM, GlcAT-S, group IVC sPLA2, GSTM5, HRSP12, IFRG15, Igkv3-4, LPLUNC3, LZIP, MRGE, Olfr827, PGAM2, PGD synthase, PGE synthase 2, PGS1, PGT, retbindin, SAA3, SAA4, SLC17A4, Slc22a27, SLC22A29, SULT1B1, SULT2A6, TPMT, TREML2, TSARG4, and Ym1.Activator of:
ABO Blood Group Antigens, AKR1C19, CRIP1, CYP2C9, H2-Q10, Lewis Blood Group Antigen, MCP-3, MNS Blood Group Antigens, MRP4, SLC22A29, and TXA synthase.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Naproxen, 1 g | sc-200506 | 1 g | $24.00 | |||
Naproxen, 5 g | sc-200506A | 5 g | $41.00 |
