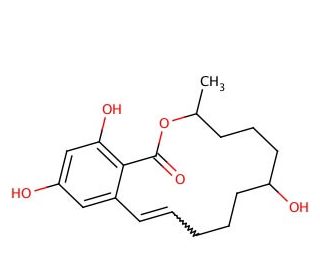
α-Zearalenol (CAS 36455-72-8)
See product citations (1)
QUICK LINKS
α-Zearalenol is a synthetic derivative of the macrolactone mycotoxin zearalenone (sc-204943) found in Fusarium roseum fungus. α-Zearalenol is estrogenically active and demonstrates induction of premature uterine growth, increase of nuclear estrogen receptor levels, and a five fold increase of ornithine decarboxylase level (ODC). The increase of ODC level was 20-fold more pronounced with α-zearalenol than zearalenone, and α-Zearalenol demonstrated three times more potent estrogenic activity than zearalenone. α-Zearalenol, a synthetic derivative of the macrolactone mycotoxin zearalenone, is a significant hepatic metabolite produced in response to the presence of zearalenone, a mycotoxin produced by fungi found in both food and animal feeds.
α-Zearalenol (CAS 36455-72-8) References
- The phytoestrogen alpha-zearalenol reverses endothelial dysfunction induced by oophorectomy in rats. | Altavilla, D., et al. 2001. Lab Invest. 81: 125-32. PMID: 11232633
- Incidence of zearalenol (Fusarium mycotoxin) in animal feed. | Mirocha, CJ., et al. 1979. Appl Environ Microbiol. 38: 749-50. PMID: 161492
- Effects of four Fusarium toxins (fumonisin B(1), alpha-zearalenol, nivalenol and deoxynivalenol) on porcine whole-blood cellular proliferation. | Luongo, D., et al. 2008. Toxicon. 52: 156-62. PMID: 18620720
- Mycotoxin alpha-zearalenol impairs the quality of preimplantation porcine embryos. | Wang, H., et al. 2012. J Reprod Dev. 58: 338-43. PMID: 22447324
- Dispositions and tissue residue of zearalenone and its metabolites α-zearalenol and β-zearalenol in broilers. | Buranatragool, K., et al. 2015. Toxicol Rep. 2: 351-356. PMID: 28962368
- Estrogenic activity of zearalenone, α-zearalenol and β-zearalenol assessed using the E-screen assay in MCF-7 cells. | Tatay, E., et al. 2018. Toxicol Mech Methods. 28: 239-242. PMID: 29057713
- Alpha-zearalenol negatively influences ram sperm parameters during liquid storage. | Abbaszadeh, S., et al. 2018. Vet Res Forum. 9: 171-178. PMID: 30065806
- Zearalenone and ß-Zearalenol But Not Their Glucosides Inhibit Heat Shock Protein 90 ATPase Activity. | Torres Acosta, JA., et al. 2019. Front Pharmacol. 10: 1160. PMID: 31680951
- Quantification of zearalenone and α-zearalenol in swine liver and reproductive tissues using GC-MS. | Pack, E., et al. 2020. Toxicon X. 8: 100058. PMID: 33089147
- Biomarkers of Exposure to Zearalenone in In Vivo and In Vitro Studies. | Llorens, P., et al. 2022. Toxins (Basel). 14: PMID: 35622538
- Identification of the naturally occurring isomer of zearalenol produced by Fusarium roseum 'Gibbosum' in rice culture. | Hagler, WM., et al. 1979. Appl Environ Microbiol. 37: 849-53. PMID: 485136
- Estrogenic activity of zearalenone and zearalanol in the neonatal rat uterus. | Sheehan, DM., et al. 1984. Teratology. 29: 383-92. PMID: 6235618
- Transformation of zearalenone and zearalenol by rat erythrocytes. | Chang, WM. and Lin, JK. 1984. Food Chem Toxicol. 22: 887-91. PMID: 6238883
- Binding properties of zearalenone mycotoxins to hepatic estrogen receptors. | Powell-Jones, W., et al. 1981. Mol Pharmacol. 20: 35-42. PMID: 6457245
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
α-Zearalenol, 5 mg | sc-202388 | 5 mg | $268.00 |
