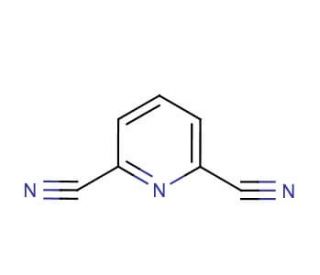
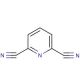
Molecular structure of 2,6-Pyridinedicarbonitrile, CAS Number: 2893-33-6
2,6-Pyridinedicarbonitrile (CAS 2893-33-6)
CAS Number:
2893-33-6
Molecular Weight:
129.12
Molecular Formula:
C7H3N3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2,6-Pyridinedicarbonitrile serves as a building block in organic synthesis. It serves as a precursor for the preparation of various heterocyclic compounds and intermediate compounds. The compound undergoes reactions such as nucleophilic substitution, condensation, and cyclization to form complex molecular structures. Its mechanism of action involves participating in chemical reactions to introduce the pyridine ring and cyano groups into the target molecules. This allows for the modification of the molecular structure and the creation of diverse chemical entities. 2,6-Pyridinedicarbonitrile plays a role in the synthesis of novel compounds with potential applications in various scientific fields.
2,6-Pyridinedicarbonitrile (CAS 2893-33-6) References
- Biotransformation of heterocyclic dinitriles by Rhodococcus erythropolis and fungal nitrilases. | Vejvoda, V., et al. 2007. Biotechnol Lett. 29: 1119-24. PMID: 17479225
- Selective aerobic oxidation of HMF to 2,5-diformylfuran on covalent triazine frameworks-supported Ru catalysts. | Artz, J., et al. 2015. ChemSusChem. 8: 672-9. PMID: 25586312
- Efficient production of hydrogen from formic acid using a covalent triazine framework supported molecular catalyst. | Bavykina, AV., et al. 2015. ChemSusChem. 8: 809-12. PMID: 25677344
- Diversity-Oriented Approach to Cyclophanes via Fischer Indolization and Ring-Closing Metathesis: Substrate-Controlled Stereochemical Outcome in RCM. | Kotha, S., et al. 2015. J Org Chem. 80: 9141-6. PMID: 26317873
- Base-Free Aqueous-Phase Oxidation of 5-Hydroxymethylfurfural over Ruthenium Catalysts Supported on Covalent Triazine Frameworks. | Artz, J. and Palkovits, R. 2015. ChemSusChem. 8: 3832-8. PMID: 26482331
- Application of Fischer Indolization under Green Conditions using Deep Eutectic Solvents. | Kotha, S. and Chakkapalli, C. 2017. Chem Rec. 17: 1039-1058. PMID: 28378920
- Facile Method for the Preparation of Covalent Triazine Framework coated Monoliths as Catalyst Support: Applications in C1 Catalysis. | Bavykina, AV., et al. 2017. ACS Appl Mater Interfaces. 9: 26060-26065. PMID: 28714671
- Single-Atom Catalysts of Precious Metals for Electrochemical Reactions. | Kim, J., et al. 2018. ChemSusChem. 11: 104-113. PMID: 28895315
- Revisiting Nitrogen Species in Covalent Triazine Frameworks. | Osadchii, DY., et al. 2017. Langmuir. 33: 14278-14285. PMID: 29182874
- Design and Synthesis of Polycycles, Heterocycles, and Macrocycles via Strategic Utilization of Ring-Closing Metathesis. | Kotha, S., et al. 2018. Chem Rec. 18: 1613-1632. PMID: 29920922
- Application of Claisen Rearrangement and Olefin Metathesis in Organic Synthesis. | Kotha, S. and Meshram, M. 2018. Chem Asian J.. PMID: 29956880
- Pyrolyzed Triazine-Based Nanoporous Frameworks Enable Electrochemical CO2 Reduction in Water. | Zhu, X., et al. 2018. ACS Appl Mater Interfaces. 10: 43588-43594. PMID: 30482016
- Recent research advancements on regioselective nitrilase: fundamental and applicative aspects. | Chen, Z., et al. 2019. Appl Microbiol Biotechnol. 103: 6393-6405. PMID: 31236614
- Exploring Soft Donor Character of the N-2-Pyrazinylmethyl Group by Coordinating Trivalent Actinides and Lanthanides Using Aminopolycarboxylates. | Grimes, TS., et al. 2020. Inorg Chem. 59: 138-150. PMID: 31430129
- Light-Intensity-Responsive Changes of Products in Photocatalytic Reduction of Nitrous Acid on a Cu-Doped Covalent Triazine Framework-TiO2 Hybrid. | Kamiya, K., et al. 2020. ChemSusChem. 13: 3462-3468. PMID: 32338454
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2,6-Pyridinedicarbonitrile, 1 g | sc-231218 | 1 g | $42.00 |
