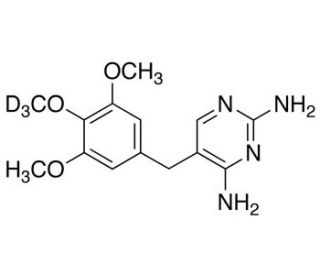
Trimethoprim-d3 (CAS 1189923-38-3)
See product citations (6)
QUICK LINKS
Trimethoprim-d3, a deuterium-labeled derivative of trimethoprim, has garnered significant interest in scientific research, particularly in pharmacokinetic and pharmacodynamic studies. Deuterium labeling involves the substitution of hydrogen atoms with deuterium, a stable isotope of hydrogen, altering the mass of the molecule while preserving its chemical properties. This modification allows for precise tracking and quantification of the compound′s distribution, metabolism, and elimination within biological systems using advanced analytical techniques such as mass spectrometry. Trimethoprim-d3 serves as a valuable tool in elucidating the pharmacokinetic parameters and bioavailability of trimethoprim, an antimicrobial agent widely used to treat bacterial infections. Researchers leverage Trimethoprim-d3 to investigate drug absorption, distribution, metabolism, and excretion (ADME) properties in both preclinical and clinical settings, providing crucial insights into its behavior and efficacy in vivo. Additionally, Trimethoprim-d3 serves as a reference standard for developing and validating analytical methods to quantify trimethoprim levels in biological samples, enhancing the accuracy and reliability of pharmacokinetic assessments. Its widespread utilization underscores its significance in advancing our understanding of antimicrobial pharmacology and optimizing strategies against bacterial pathogens through rigorous scientific research.
Trimethoprim-d3 (CAS 1189923-38-3) References
- Simultaneous detection of antibiotics and other drug residues in the dissolved and particulate phases of water by an off-line SPE combined with on-line SPE-LC-MS/MS: Method development and application. | Tlili, I., et al. 2016. Sci Total Environ. 563-564: 424-33. PMID: 27151499
- Determination of 21 antibiotics in sea cucumber using accelerated solvent extraction with in-cell clean-up coupled to ultra-performance liquid chromatography-tandem mass spectrometry. | Zhu, M., et al. 2018. Food Chem. 258: 87-94. PMID: 29655759
- Ammonia Monooxygenase-Mediated Cometabolic Biotransformation and Hydroxylamine-Mediated Abiotic Transformation of Micropollutants in an AOB/NOB Coculture. | Yu, Y., et al. 2018. Environ Sci Technol. 52: 9196-9205. PMID: 30004677
- Simultaneous determination of multiclass antibiotics and their metabolites in four types of field-grown vegetables. | Tadić, Đ., et al. 2019. Anal Bioanal Chem. 411: 5209-5222. PMID: 31183522
- Multiantibiotic residues in commercial fish from Argentina. The presence of mixtures of antibiotics in edible fish, a challenge to health risk assessment. | Griboff, J., et al. 2020. Food Chem. 332: 127380. PMID: 32603916
- Analysis of pharmaceuticals in fish using ultrasound extraction and dispersive spe clean-up on que Z-Sep/C18 followed by LC-QToF-MS detection. | Peña-Herrera, JM., et al. 2020. MethodsX. 7: 101010. PMID: 32793428
- Emerging contaminants detected in aquaculture sites in Java, Indonesia. | Hidayati, NV., et al. 2021. Sci Total Environ. 773: 145057. PMID: 33592457
- A HPLC-MS/MS method for screening of selected antibiotic adulterants in herbal drugs. | Mwankuna, CJ., et al. 2022. Anal Methods. 14: 1060-1068. PMID: 35195137
- The assessment of environmental risk related to the occurrence of pharmaceuticals in bottom sediments of the Odra River estuary (SW Baltic Sea). | Kucharski, D., et al. 2022. Sci Total Environ. 828: 154446. PMID: 35283119
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trimethoprim-d3, 1 mg | sc-220337 | 1 mg | $480.00 |
