Lithium tetraborate (CAS 12007-60-2)
QUICK LINKS
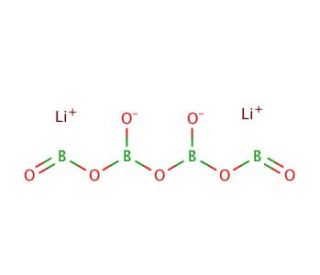
Lithium tetraborate, with the chemical formula Li2B4O7 and CAS number 12007-60-2, is an inorganic compound that is particularly valued in analytical chemistry and materials science due to its unique chemical properties. This white, crystalline compound acts as a powerful flux in the fusion technique for preparing glassy or molten samples for spectroscopic analysis. In this context, lithium tetraborate is used to lower the melting points of substances, thereby facilitating the dissolution of oxidic materials into a homogenous molten mass. This is crucial for the accurate determination of the elemental composition of various materials using techniques such as X-ray fluorescence (XRF) and inductively coupled plasma (ICP) spectroscopy. The ability to create a uniform sample matrix without any mineralogical effects allows for high precision in these analytical methods. Additionally, lithium tetraborate finds applications in the synthesis of special glass types, where its presence influences the properties of the glass, such as increasing the chemical durability and lowering the viscosity. These characteristics are essential for producing glasses with specific optical and thermal properties. Moreover, its role in the geochemical analysis of rocks and minerals makes it an indispensable tool in geology research, contributing to the understanding of the Earth′s crust and mantle processes.
Lithium tetraborate (CAS 12007-60-2) References
- Interaction between a Martian Regolith Simulant and Fungal Organic Acids in the Biomining Perspective. | Vezzola, M., et al. 2023. J Fungi (Basel). 9: PMID: 37888232
- Red clays indicate sub-aerial exposure of the Rio Grande Rise during the Eocene volcanic episode. | Srivastava, P., et al. 2023. Sci Rep. 13: 19092. PMID: 37925541
- Local Structure and Speciation-Driven UO22+ → Sm3+ Energy Transfer for Enhanced Luminescence in Li2B4O7. | Balhara, A., et al. 2023. Inorg Chem. 62: 20258-20270. PMID: 38033302
- Research on improving the accuracy of laser-induced breakdown spectroscopy analysis by considering plasma attenuation rate characteristics. | Li, C., et al. 2024. Anal Chim Acta. 1287: 342065. PMID: 38182372
- Dissolution behavior of stone wool fibers in synthetic lung fluids: Impact of iron oxidation state changes induced by heat treatment for binder removal. | Okhrimenko, DV., et al. 2024. Toxicol Lett. 393: 33-46. PMID: 38232781
- Season-dependent physiological behavior of Miscanthus x giganteus growing on heavy-metal contaminated areas in relation to soil properties. | Lutts, S., et al. 2024. Heliyon. 10: e25943. PMID: 38384526
- In vitro ruminal fermentation, core nutrient, fatty acids and mineral matter of pennyroyal (Mentha pulegium L.) herbage at different phenological stages. | Kara, K., et al. 2024. Vet Med Sci. 10: e1397. PMID: 38450960
- New structural-optical effect in LiF-Li2B4O7 thin-film structures in the crystallization. | Maslyuk, V., et al. 2024. Luminescence. 39: e4733. PMID: 38556359
- Structural and thermoluminescence properties of lithium borate glass matrices under UV and beta radiation. | Menezes, DLA., et al. 2024. Luminescence. 39: e4732. PMID: 38556715
- Enhancing iron biogeochemical cycling for canga ecosystem restoration: insights from microbial stimuli. | da Silva, RDSS., et al. 2024. Front Microbiol. 15: 1352792. PMID: 38827154
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Lithium tetraborate, 1 kg | sc-203113 | 1 kg | $291.00 |
