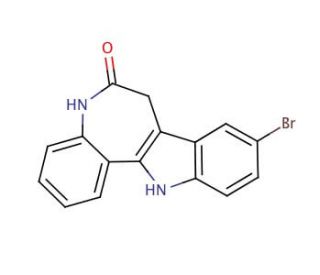
Molecular structure of Kenpaullone, CAS Number: 142273-20-9
Kenpaullone (CAS 142273-20-9)
See product citations (1)
Alternate Names:
9-Bromo-7,12-dihydroindolo-[3,2-d][1]benzazepin-6(5H)-one;NSC 664704
Application:
Kenpaullone is a Cdk and GKS-3 inhibitor
CAS Number:
142273-20-9
Purity:
≥98%
Molecular Weight:
327.17
Molecular Formula:
C16H11BrN2O
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Kenpaullone is an ATP-competitive inhibitor of several CDKs (cyclin-dependent kinases) as well as GSK-3beta (glycogen synthase kinase 3beta). It may act to inhibit GSK-3 with an IC50 value of 0.23 muM and CDK1/cyclin B, CDK2/cyclin A, CDK5/p25, and lymphocyte kinase with IC50 values of 0.4, 0.68, 0.85, and 0.47 muM, respectively. Due to the compound′s activity, Kenpaullone is useful in the study of cell cycle regulators. Kenpaullone is an inhibitor of cyclin E, ERK 2 and p35.
Kenpaullone (CAS 142273-20-9) References
- Discovery and initial characterization of the paullones, a novel class of small-molecule inhibitors of cyclin-dependent kinases. | Zaharevitz, DW., et al. 1999. Cancer Res. 59: 2566-9. PMID: 10363974
- Indirubins inhibit glycogen synthase kinase-3 beta and CDK5/p25, two protein kinases involved in abnormal tau phosphorylation in Alzheimer's disease. A property common to most cyclin-dependent kinase inhibitors? | Leclerc, S., et al. 2001. J Biol Chem. 276: 251-60. PMID: 11013232
- The specificities of protein kinase inhibitors: an update. | Bain, J., et al. 2003. Biochem J. 371: 199-204. PMID: 12534346
- New thiophene analogues of kenpaullone: synthesis and biological evaluation in breast cancer cells. | Brault, L., et al. 2005. Eur J Med Chem. 40: 757-63. PMID: 16122578
- Small molecule GSK-3 inhibitors increase neurogenesis of human neural progenitor cells. | Lange, C., et al. 2011. Neurosci Lett. 488: 36-40. PMID: 21056624
- CDK inhibitors suppress Th17 and promote iTreg differentiation, and ameliorate experimental autoimmune encephalomyelitis in mice. | Yoshida, H., et al. 2013. Biochem Biophys Res Commun. 435: 378-84. PMID: 23665028
- Inhibition of CDK2 promotes inducible regulatory T-cell differentiation through TGFβ-Smad3 signaling pathway. | Gu, H., et al. 2014. Cell Immunol. 290: 138-44. PMID: 24978614
- The inhibitors of cyclin-dependent kinases and GSK-3β enhance osteoclastogenesis. | Akiba, Y., et al. 2016. Biochem Biophys Rep. 5: 253-258. PMID: 28955831
- Developmental effects of the protein kinase inhibitor kenpaullone on the sea urchin embryo. | Anello, L., et al. 2018. Comp Biochem Physiol C Toxicol Pharmacol. 204: 36-44. PMID: 29128602
- Protective effects of kenpaullone on cardiomyocytes following H2O2-induced oxidative stress are attributed to inhibition of connexin 43 degradation by SGSM3. | Joo, HC., et al. 2018. Biochem Biophys Res Commun. 499: 368-373. PMID: 29577900
- Identification of GSK3β inhibitor kenpaullone as a temozolomide enhancer against glioblastoma. | Kitabayashi, T., et al. 2019. Sci Rep. 9: 10049. PMID: 31296906
- KLF4 inhibition promotes the expansion of keratinocyte precursors from adult human skin and of embryonic-stem-cell-derived keratinocytes. | Fortunel, NO., et al. 2019. Nat Biomed Eng. 3: 985-997. PMID: 31636412
- KLF4 inhibition by Kenpaullone induces cytotoxicity and chemo sensitization in B-NHL cell lines via YY1 independent. | Montecillo-Aguado, M., et al. 2021. Leuk Lymphoma. 62: 1422-1431. PMID: 33410342
Inhibitor of:
2410141K09Rik, 4930525M21Rik, 4933439F11Rik, Alsin, ANKRD20A1, ANKRD20A3, atrophin-1, ATXN7L1, Bex3, BTF3, BTG3, C6orf145, CDCrel-1, Cdk1, Cdk2, Cdk5, CITED4, COASTER, CPAN, cyclin A, cyclin B, cyclin C, cyclin E, D14Ertd449e, DBX2, DEPDC1, DIEXF, EG668381, ERK 2, Exendin 4, FAM160A1, FEV, GKLF, Gm355, GRIF-1, GSKIP, Gtlf3b, HBP1, hCG_1988300, JDP2, Krüppel-like factor (KLF), Lambda 5, LOC100041193, LOC100041397, LOC728222, LOC728478, LOC729504, LOC729991-MEF2B, MYL9, NanogNB, nArgBP2, NEGR1, NPB, OLIG1, p35, Paip2, PEBP2αA, PITSLRE A, PSP, Repetin, Ser/Thr Protein Kinase, Speedy E4, STELLA, TCEAL1, TCHHL1, TLK2, TSGA10IP, TTBK2, WBSCR11, X11γ, YAP, ZBTB22, ZNF2, ZNF295, ZNF449, and ZNF85.Activator of:
4931431F19Rik, C16orf87_4921524J17Rik, C17orf47, casein kinase Iγ1, Cdc45, CDKL5, Clik1, CLK1, CLK3, Dyrk4, IFT46, LIT-1, Mat1, Neurotrypsin, p39, PISSLRE, TNIK, TTBK2, and VPS45.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Kenpaullone, 1 mg | sc-200643 | 1 mg | $61.00 | |||
Kenpaullone, 5 mg | sc-200643A | 5 mg | $153.00 | |||
Kenpaullone, 10 mg | sc-200643B | 10 mg | $231.00 | |||
Kenpaullone, 25 mg | sc-200643C | 25 mg | $505.00 |
