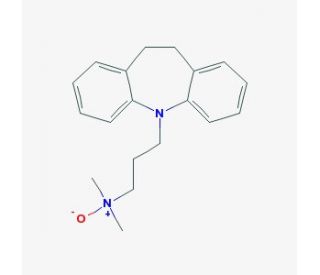
Molecular structure of Imipramine N-oxide, CAS Number: 6829-98-7
Imipramine N-oxide (CAS 6829-98-7)
Alternate Names:
Imipraminoxide
Application:
Imipramine N-oxide is a tertiary amine-oxidized metabolite of Imipramine
CAS Number:
6829-98-7
Purity:
98%
Molecular Weight:
296.4
Molecular Formula:
C19H24N2O
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Imipramine N-oxide is the tertiary amine-oxidized form and a natural metabolite of the tricyclic serotonin/dopamine transport inhibitor Imipramine (sc-207753). FMO (Flavin-containing monooxygenase) enzymes are responsible for the oxidation of nucleophilic heteroatoms such as the tertiary amine of Imipramine, utilizing the bound cofactor flavin for redox coupling. Imipramine N-oxide can be used as a standard for investigating the presence and activity of FMO enzymes in relation to the FMO-mediated oxidation of Imipramine. Imipramine N-oxide is a substrate of FMO.
Imipramine N-oxide (CAS 6829-98-7) References
- Biphasic kinetics of imipramine N-oxidation in rat brain microsomes. | Narimatsu, S., et al. 1999. Biol Pharm Bull. 22: 253-6. PMID: 10220279
- Oxidation of tamoxifen by human flavin-containing monooxygenase (FMO) 1 and FMO3 to tamoxifen-N-oxide and its novel reduction back to tamoxifen by human cytochromes P450 and hemoglobin. | Parte, P. and Kupfer, D. 2005. Drug Metab Dispos. 33: 1446-52. PMID: 15987777
- N-demethylation and N-oxidation of imipramine in rat thoracic aortic endothelial cells. | Ueda, Y., et al. 2014. In Vitro Cell Dev Biol Anim. 50: 496-501. PMID: 24647745
- Metabolism of drugs in the eye. Menadione-dependent reduction of tertiary amine N-oxide by preparations from bovine ocular tissues. | Shimada, S., et al. 1989. Curr Eye Res. 8: 1309-13. PMID: 2627798
- What is the contribution of human FMO3 in the N-oxygenation of selected therapeutic drugs and drugs of abuse? | Wagmann, L., et al. 2016. Toxicol Lett. 258: 55-70. PMID: 27320963
- Aldehyde oxidase and its role as a drug metabolizing enzyme. | Dalvie, D. and Di, L. 2019. Pharmacol Ther. 201: 137-180. PMID: 31128989
- Metabolism by Aldehyde Oxidase: Drug Design and Complementary Approaches to Challenges in Drug Discovery. | Manevski, N., et al. 2019. J Med Chem. 62: 10955-10994. PMID: 31385704
- Aldehyde oxidase at the crossroad of metabolism and preclinical screening. | Cheshmazar, N., et al. 2019. Drug Metab Rev. 51: 428-452. PMID: 31549868
- Population Pharmacokinetics of Sulindac and Genetic Polymorphisms of FMO3 and AOX1 in Women with Preterm Labor. | Sung, JW., et al. 2020. Pharm Res. 37: 44. PMID: 31993760
- Metabolic N-Dealkylation and N-Oxidation as Elucidators of the Role of Alkylamino Moieties in Drugs Acting at Various Receptors. | Eh-Haj, BM. 2021. Molecules. 26: PMID: 33805491
- Non-P450 Drug-Metabolizing Enzymes: Contribution to Drug Disposition, Toxicity, and Development. | Fukami, T., et al. 2022. Annu Rev Pharmacol Toxicol. 62: 405-425. PMID: 34499522
- Differences between water-soluble and water-insoluble melanin derived from Inonotus hispidus mushroom. | Li, X., et al. 2022. Food Chem X. 16: 100498. PMID: 36519091
- Imipramine metabolites in blood of patients during therapy and after overdose. | Gram, LF., et al. 1983. Clin Pharmacol Ther. 33: 335-42. PMID: 6825388
- Further characterization of rat brain flavin-containing monooxygenase. Metabolism of imipramine to its N-oxide. | Bhagwat, SV., et al. 1996. Biochem Pharmacol. 51: 1469-75. PMID: 8630088
Substrate of:
FMO.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Imipramine N-oxide, 5 mg | sc-200973 | 5 mg | $96.00 | |||
Imipramine N-oxide, 25 mg | sc-200973A | 25 mg | $360.00 |
