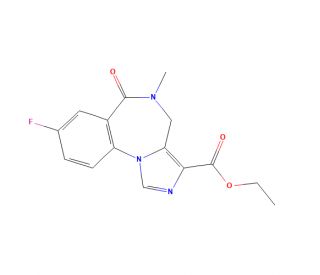
Flumazenil (Ro 15-1788) (CAS 78755-81-4)
See product citations (10)
QUICK LINKS
Flumazenil (Ro 15-1788) is an antagonist to benzodiazepine allosteric binding on the GABAA receptor. Administration of Flumazenil (Ro 15-1788) may act to reverse many of the physiological effects associated with benzodiazepines and their modification of the GABAA system. Flumazenil (Ro 15-1788) has demonstrated the ability to regulate the activity of important neurotransmitters, including GABA, serotonin, and glutamate. In laboratory applications, Flumazenil (Ro 15-1788) serves as useful for investigating the impact of benzodiazepines on various cellular mechanisms. It enables the study of benzodiazepine effects on gene expression, protein synthesis, and intricate cell signaling pathways.
Flumazenil (Ro 15-1788) (CAS 78755-81-4) References
- Molecular and neurochemical evaluation of the effects of etizolam on GABAA receptors under normal and stress conditions. | Sanna, E., et al. 1999. Arzneimittelforschung. 49: 88-95. PMID: 10083975
- Cerebral blood perfusion after treatment with zolpidem and flumazenil in the baboon. | Clauss, RP., et al. 2002. Arzneimittelforschung. 52: 740-4. PMID: 12442636
- Flumazenil. A reappraisal of its pharmacological properties and therapeutic efficacy as a benzodiazepine antagonist. | Brogden, RN. and Goa, KL. 1991. Drugs. 42: 1061-89. PMID: 1724638
- Comparative analysis of pharmaceuticals versus industrial chemicals acute aquatic toxicity classification according to the United Nations classification system for chemicals. Assessment of the (Q)SAR predictability of pharmaceuticals acute aquatic toxicity and their predominant acute toxic mode-of-action. | Sanderson, H. and Thomsen, M. 2009. Toxicol Lett. 187: 84-93. PMID: 19429249
- Reduction of reticulata neuronal activity by zolpidem and alpidem, two imidazopyridines with high affinity for type I benzodiazepine receptors. | Mereu, G., et al. 1990. Eur J Pharmacol. 179: 339-45. PMID: 1973107
- The stability of methyl-, ethyl- and fluoroethylesters against carboxylesterases in vitro: there is no difference. | Nics, L., et al. 2011. Nucl Med Biol. 38: 13-7. PMID: 21220125
- Validation of a high throughput flow cytometric in vitro micronucleus assay including assessment of metabolic activation in TK6 cells. | Thougaard, AV., et al. 2014. Environ Mol Mutagen. 55: 704-18. PMID: 25111698
- α-Pinene, a Major Constituent of Pine Tree Oils, Enhances Non-Rapid Eye Movement Sleep in Mice through GABAA-benzodiazepine Receptors. | Yang, H., et al. 2016. Mol Pharmacol. 90: 530-539. PMID: 27573669
- Chronic treatment with Ro 15-1788 distinguishes between its benzodiazepine antagonist, agonist and inverse agonist properties. | File, SE., et al. 1986. Psychopharmacology (Berl). 89: 113-7. PMID: 3090585
- Source identity shapes spatial preference in primary auditory cortex during active navigation. | Amaro, D., et al. 2021. Curr Biol. 31: 3875-3883.e5. PMID: 34192513
- Effects of flumazenil on ethanol withdrawal syndrome in rats. | Uzbay, IT., et al. 1995. Arzneimittelforschung. 45: 120-4. PMID: 7710430
- Flumazenil: an antidote for benzodiazepine toxicity. | Krisanda, TJ. 1993. Am Fam Physician. 47: 891-5. PMID: 8438687
- Effect of neuroactive steroids on [3H]flumazenil binding to the GABAA receptor complex in vitro. | Bertz, RJ., et al. 1995. Neuropharmacology. 34: 1169-75. PMID: 8532187
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Flumazenil (Ro 15-1788), 25 mg | sc-200161 | 25 mg | $110.00 | |||
Flumazenil (Ro 15-1788), 100 mg | sc-200161A | 100 mg | $370.00 |
