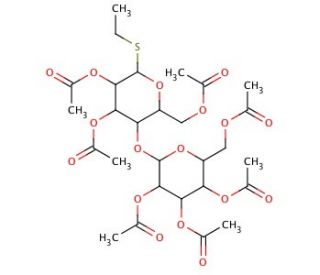
Ethyl 2,3,6,2′,3′,4′,6′-Hepta-O-acetyl-α,β-D-thiolactopyranoside
QUICK LINKS
Ethyl 2,3,6,2′,3′,4′,6′-Hepta-O-acetyl-α,β-D-thiolactopyranoside is a compound widely used in carbohydrate chemistry research due to its unique structural features and diverse synthetic applications. This chemical serves as a versatile building block for the synthesis of complex carbohydrate derivatives, owing to its ability to undergo selective deprotection and functionalization reactions. Mechanistically, the hepta-O-acetyl groups provide protection to the hydroxyl groups of the sugar moiety, allowing for precise control over chemical modifications. Researchers have employed this compound in the synthesis of various glycoconjugates, glycomimetics, and glycopeptides for investigating carbohydrate-protein interactions, glycobiology, and chemical biology. Additionally, it has been utilized in the development of carbohydrate-based materials, such as glycoarrays and glycosylated nanoparticles, for applications in drug delivery, diagnostics, and biomaterials science. Furthermore, its use in chemical synthesis has enabled the preparation of structurally diverse carbohydrate derivatives with tailored properties for studying structure-function relationships and designing novel carbohydrate-based research tools and materials. Overall, Ethyl 2,3,6,2′,3′,4′,6′-Hepta-O-acetyl-α,β-D-thiolactopyranoside represents a valuable tool in carbohydrate research, offering opportunities for advancing our understanding of carbohydrate biology and developing innovative applications in various scientific disciplines.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl 2,3,6,2',3',4',6'-Hepta-O-acetyl-α,β-D-thiolactopyranoside, 1 g | sc-218369 | 1 g | $360.00 |
