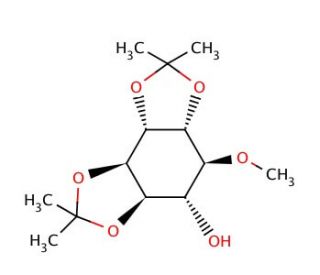
1,2:5,6-Di-O-isopropylidene-D-chiro-inositol (CAS 57819-56-4)
QUICK LINKS
1,2:5,6-Di-O-isopropylidene-D-chiro-inositol, commonly referred to as DICI, has garnered significant attention in scientific research due to its pivotal role in elucidating the functions of inositol derivatives in cellular processes. As a derivative of chiro-inositol, DICI serves as a valuable tool in investigating the mechanisms underlying inositol signaling pathways and their implications in cellular physiology. One prominent mechanism of action of DICI involves its ability to mimic the structural features of chiro-inositol, thereby modulating inositol-dependent signaling cascades. Research indicates that DICI can competitively bind to inositol-binding proteins, thereby influencing downstream signaling events mediated by these proteins. Moreover, DICI has been instrumental in studying the physiological roles of inositol derivatives in diverse cellular processes, including cell growth, differentiation, and intracellular signaling. Additionally, DICI has been utilized as a chemical probe in structural studies aimed at elucidating the binding interactions between inositol derivatives and their target proteins, providing valuable insights into the molecular mechanisms underlying inositol signaling. Overall, DICI represents a valuable asset in biochemical research, offering opportunities to unravel the complex roles of inositol derivatives in cellular physiology and pathology.
1,2:5,6-Di-O-isopropylidene-D-chiro-inositol (CAS 57819-56-4) References
- Chiral synthesis of D- and L-myo-inositol 1,4,5-trisphosphate. | Tegge, W. and Ballou, CE. 1989. Proc Natl Acad Sci U S A. 86: 94-8. PMID: 2783488
- Ruthenium complexes that incorporate a chiro-inositol derived diphosphinite ligand as catalysts for asymmetric hydrogenation reactions[J]. | Falshaw A, Gainsford G J, Lensink C. 2007,. Polyhedron,. 26(2):: 329-337.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2:5,6-Di-O-isopropylidene-D-chiro-inositol, 1 g | sc-222905 | 1 g | $144.00 |
