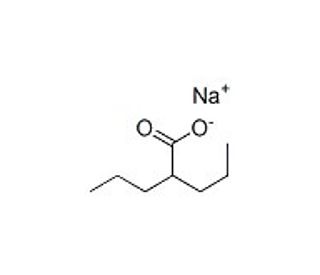
Valproic acid sodium salt (CAS 1069-66-5)
See product citations (9)
QUICK LINKS
Valproic Acid, Sodium Salt is a branched chain fatty acid which is reported to inhance central GABAergic neurotransmission and inhibit Sodium Channel Protein (Na+ channels). This compound is also reported to cause inositol depletion, activate the ERK pathway, inhibit GSK-3alpha and GSK-3beta. Valproic Acid has been reported to be a potent inhibitor of HDAC (histone deacetylase) in vitro (IC50 = 400 muM for HDAC1), thereby relieving HDAC-dependent transcriptional repression and causes the hyperacetylation of histones in cultured cells. In animal studies, Valproic Acid has been observed to reduce tumor growth and metastasis formation. Additionally, Valproic Acid is reported to activate Wnt-dependent gene expression and to mimic trichostatin A (sc-3511) in the inhibition of histone deacetylase. This compound is also an inhibitor of the CYP2C9 enzyme. Metabolites of Valproic Acid are also available as: Valproic Acid (sc-213144) 3-Hydroxy Valproic Acid (sc-209599) 3-Keto Valproic Acid Sodium Salt (sc-216476) 4-Hydroxy Valproic Acid Sodium Salt (Mixture of Diastereomers) (sc-216870) rac 5-Hydroxy Valproic Acid Sodium Salt (sc-208266) Valproic Acid beta-D-Glucuronide Allyl Ester (sc-220360) Valproic Acid beta-D-Glucuronide (sc-213145) Labelled forms of Valproic Acid are available as: Valproic Acid-d6 (sc-213146) Valproic Acid-d6 beta-D-Glucuronide (sc-220361) rac 5-Hydroxy Valproic Acid-d7 Sodium Salt (sc-219789)
Valproic acid sodium salt (CAS 1069-66-5) References
- Actions of sodium valproate on the central nervous system. | Tunnicliff, G. 1999. J Physiol Pharmacol. 50: 347-65. PMID: 10574466
- Histone deacetylase is a direct target of valproic acid, a potent anticonvulsant, mood stabilizer, and teratogen. | Phiel, CJ., et al. 2001. J Biol Chem. 276: 36734-41. PMID: 11473107
- In vitro evaluation of valproic acid as an inhibitor of human cytochrome P450 isoforms: preferential inhibition of cytochrome P450 2C9 (CYP2C9). | Wen, X., et al. 2001. Br J Clin Pharmacol. 52: 547-53. PMID: 11736863
- Valproic acid defines a novel class of HDAC inhibitors inducing differentiation of transformed cells. | Göttlicher, M., et al. 2001. EMBO J. 20: 6969-78. PMID: 11742974
- Valproic acid induces up- or down-regulation of gene expression responsible for the neuronal excitation and inhibition in rat cortical neurons through its epigenetic actions. | Fukuchi, M., et al. 2009. Neurosci Res. 65: 35-43. PMID: 19463867
- Valproic acid-associated vanishing bile duct syndrome. | Gökçe, S., et al. 2010. J Child Neurol. 25: 909-11. PMID: 20388938
- In vitro interactions of amantadine hydrochloride, R-(-)-deprenyl hydrochloride and valproic acid sodium salt with antifungal agents against filamentous fungal species causing central nervous system infection. | Galgóczy, L., et al. 2012. Acta Biol Hung. 63: 490-500. PMID: 23134606
- Combined prenatal and postnatal butyl paraben exposure produces autism-like symptoms in offspring: comparison with valproic acid autistic model. | Ali, EH. and Elgoly, AH. 2013. Pharmacol Biochem Behav. 111: 102-10. PMID: 24036472
- Decreased mTOR signaling pathway in human idiopathic autism and in rats exposed to valproic acid. | Nicolini, C., et al. 2015. Acta Neuropathol Commun. 3: 3. PMID: 25627160
- A combination of valproic acid sodium salt, CHIR99021, E-616452, tranylcypromine, and 3-Deazaneplanocin A causes stem cell-like characteristics in cancer cells. | Sha, S., et al. 2017. Oncotarget. 8: 53302-53312. PMID: 28881812
- Crystal engineering of valproic acid and carbamazepine to improve hygroscopicity and dissolution profile. | Khajir, S., et al. 2021. Drug Dev Ind Pharm. 47: 1674-1679. PMID: 35196936
- Evaluation of mitochondrial oxidative toxicity in mammalian cardiomyocytes by determining the highly reproducible and reliable increase in mitochondrial superoxides after exposure to therapeutic drugs. | Ahn, D., et al. 2022. Toxicol In Vitro. 83: 105393. PMID: 35618243
- Modelling of the pharmacodynamic interaction between phenytoin and sodium valproate. | Della Paschoa, OE., et al. 1998. Br J Pharmacol. 125: 1610-6. PMID: 9884091
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Valproic acid sodium salt, 1 g | sc-202378A | 1 g | $21.00 | |||
Valproic acid sodium salt, 5 g | sc-202378 | 5 g | $37.00 | |||
Valproic acid sodium salt, 25 g | sc-202378B | 25 g | $131.00 | |||
Valproic acid sodium salt, 100 g | sc-202378C | 100 g | $369.00 |
