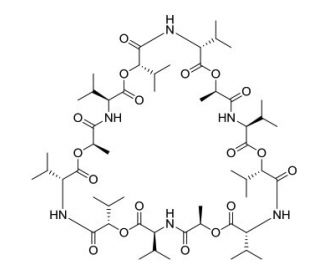
Valinomycin (CAS 2001-95-8)
See product citations (3)
QUICK LINKS
Valinomycin is a naturally occurring dodecadepsipeptide known for its ability to transport potassium ions across lipid membranes. It is used extensively in research studies that investigate ion transport and membrane permeability. By forming a complex with potassium ions, valinomycin effectively creates a pathway for these ions to move across cell membranes, a process that is otherwise highly regulated by the cell. This ionophoric action is utilized in studies of mitochondrial function and bioenergetics, as valinomycin can disrupt the electrochemical gradient by increasing membrane permeability to potassium. In addition, researchers use valinomycin to understand the mechanisms behind ion selectivity and channel-forming properties of peptides and other macromolecules. The compound is also instrumental in synthetic membrane experiments, which are designed to mimic and study the properties of biological membranes.
Valinomycin (CAS 2001-95-8) References
- Induction of apoptosis by valinomycin: mitochondrial permeability transition causes intracellular acidification. | Furlong, IJ., et al. 1998. Cell Death Differ. 5: 214-21. PMID: 10200467
- Permeability transition-independent release of mitochondrial cytochrome c induced by valinomycin. | Shinohara, Y., et al. 2002. Eur J Biochem. 269: 5224-30. PMID: 12392554
- Involvement of a Na+/HCO-3 cotransporter in mouse sperm capacitation. | Demarco, IA., et al. 2003. J Biol Chem. 278: 7001-9. PMID: 12496293
- Cyclosporin A does not protect the disruption of the inner mitochondrial membrane potential induced by potassium ionophores in intact K562 cells. | Marques-Santos, LF., et al. 2006. Cell Biol Int. 30: 197-204. PMID: 16376584
- Valinomycin-induced apoptosis in Chinese hamster ovary cells. | Abdalah, R., et al. 2006. Neurosci Lett. 405: 68-73. PMID: 16857314
- Comparative analysis of apoptotic pathways in rat, mouse, and hamster spermatozoa. | Cisternas, P. and Moreno, RD. 2006. Mol Reprod Dev. 73: 1318-25. PMID: 16868928
- Permissive role of calcium on regulatory volume decrease in freshly isolated mouse cholangiocytes. | Park, JS., et al. 2007. Pflugers Arch. 455: 261-71. PMID: 17503070
- Cereulide and valinomycin, two important natural dodecadepsipeptides with ionophoretic activities. | Kroteń, MA., et al. 2010. Pol J Microbiol. 59: 3-10. PMID: 20568524
- Effects and mechanisms of action of ionophorous antibiotics valinomycin and salinomycin-Na on Babesia gibsoni in vitro. | Yamasaki, M., et al. 2009. J Parasitol. 95: 1532-8. PMID: 20929429
- Perturbation of intracellular K(+) homeostasis with valinomycin promotes cell death by mitochondrial swelling and autophagic processes. | Klein, B., et al. 2011. Apoptosis. 16: 1101-17. PMID: 21877215
- Mitochondrial uncouplers inhibit hepatic stellate cell activation. | Guimarães, EL., et al. 2012. BMC Gastroenterol. 12: 68. PMID: 22686625
- The voltage-gated proton channel Hv1/VSOP inhibits neutrophil granule release. | Okochi, Y., et al. 2016. J Leukoc Biol. 99: 7-19. PMID: 25990245
- Study of mitophagy and ATP-related metabolomics based on β-amyloid levels in Alzheimer's disease. | Xiong, X., et al. 2020. Exp Cell Res. 396: 112266. PMID: 32905804
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Valinomycin, 25 mg | sc-200991 | 25 mg | $250.00 |
