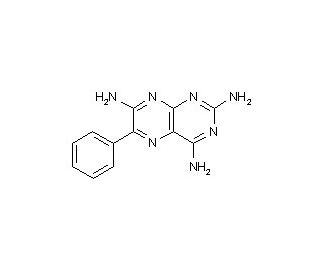
Triamterene (CAS 396-01-0)
QUICK LINKS
Triamterene is a synthetic compound that is widely studied in the context of renal physiology and electrolyte balance research. It is recognized for its function as a potassium-sparing diuretic in studies that do not involve clinical applications. Researchers focus on triamterene′s ability to inhibit the epithelial sodium channels in the distal renal tubules, which is a mechanism of interest in the regulation of sodium reabsorption and potassium excretion. In cellular and molecular biology, triamterene is used to investigate the modulation of ion transport processes and their impact on cellular homeostasis. Additionally, its role in the study of membrane transporters and the effect on transepithelial ion flux is of particular relevance in understanding the pathways that maintain electrolyte balance. Triamterene also serves as a tool in evaluating the function of nephron segments under various experimental conditions.
Triamterene (CAS 396-01-0) References
- Triamterene, a new diuretic drug. II. Clinical trial in oedematous patients. | BABA, WI., et al. 1962. Br Med J. 2: 760-4. PMID: 13863614
- Triamterene, a new natruretic agent. Preliminary observations in man. | CROSLEY, AP., et al. 1962. Ann Intern Med. 56: 241-51. PMID: 13882367
- Triamterene-beta-cyclodextrin systems: preparation, characterization and in vivo evaluation. | Mukne, AP. and Nagarsenker, MS. 2004. AAPS PharmSciTech. 5: E19. PMID: 15198540
- The delivery of triamterene by cucurbit[7]uril: synthesis, structures and pharmacokinetics study. | Ma, WJ., et al. 2013. Mol Pharm. 10: 4698-705. PMID: 24188081
- Triamterene in the Treatment of Hypertension: More Than Just Potassium Sparing? | Smetana, GW. 2016. J Gen Intern Med. 31: 7-8. PMID: 26385116
- Triamterene and renal stone formation: the influence of triamterene and triamterene stones on calcium oxalate crystallization. | White, DJ. and Nancollas, GH. 1987. Calcif Tissue Int. 40: 79-84. PMID: 3105837
- Preparation and characterization of triamterene complex with ascorbic acid derivatives. | Onoda, H., et al. 2020. Drug Dev Ind Pharm. 46: 2032-2040. PMID: 33104388
- Triamterene induces autophagic degradation of lysosome by exacerbating lysosomal integrity. | Park, NY., et al. 2021. Arch Pharm Res. 44: 621-631. PMID: 34100261
- Triamterene bladder calculus. | Hollander, JB. 1987. Urology. 30: 154-5. PMID: 3617299
- Triamterene stones and computerized axial tomography. | Guevara, A., et al. 1986. Urology. 27: 104-6. PMID: 3946031
- Triamterene and renal lithiasis: a review. | Carey, RA., et al. 1984. Clin Ther. 6: 302-9. PMID: 6373008
- Triamterene and renal stone formation. | White, DJ. and Nancollas, GH. 1982. J Urol. 127: 593-7. PMID: 7062445
- Pharmacokinetics of triamterene and its metabolite in man. | Hasegawa, J., et al. 1982. J Pharmacokinet Biopharm. 10: 507-23. PMID: 7166735
- Triamterene inhibits the delayed rectifier potassium current (IK) in guinea pig ventricular myocytes. | Daleau, P. and Turgeon, J. 1994. Circ Res. 74: 1114-20. PMID: 8187277
- Potassium and magnesium retaining triamterene derivatives. | Priewer, H. and Ullrich, F. 1997. Pharmazie. 52: 179-81. PMID: 9109165
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Triamterene, 1 g | sc-213103A | 1 g | $22.00 | |||
Triamterene, 5 g | sc-213103 | 5 g | $54.00 |
