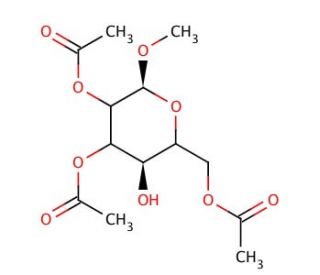
Methyl 2,3,6-Tri-O-acetyl-α-D-glucopyranoside
QUICK LINKS
Methyl 2,3,6-Tri-O-acetyl-α-D-glucopyranoside is a derivative of glucose that is extensively used in synthetic carbohydrate chemistry for its role as a glycosyl donor in glycosylation reactions. This molecule modifies the basic glucose structure by acetylating the hydroxyl groups at the 2, 3, and 6 positions, which not only protects these groups during chemical reactions but also enhances the reactivity of the anomeric center. This enhancement makes the compound an effective tool in the synthesis of more complex oligosaccharides and glycoconjugates. In research, this compound′s primary utility lies in its ability to participate in selective glycosylation reactions, where it acts as a controlled source of glucose units. These reactions are crucial for constructing glycosidic linkages with precise stereochemical control, which is essential for the synthesis of natural and unnatural glycosides. The acetyl groups can be removed under mild conditions, allowing the unveiling of the native hydroxyl groups for further functionalization or for studies on their natural interactions. Through its use, Methyl 2,3,6-Tri-O-acetyl-α-D-glucopyranoside provides significant insights into the mechanisms of glycosylation, helping to advance our understanding of how carbohydrates are assembled in a laboratory setting. This knowledge is vital for applications in materials science, biotechnology, and fundamental carbohydrate research, pushing the boundaries of synthetic chemistry and glycoscience.
Methyl 2,3,6-Tri-O-acetyl-α-D-glucopyranoside References
- Stereocontrolled synthesis of D- and L-beta-rhamnopyranosides with 4-O-6-S-alpha-cyanobenzylidene-protected 6-thiorhamnopyranosyl thioglycosides. | Crich, D. and Li, L. 2009. J Org Chem. 74: 773-81. PMID: 19132946
- Screening of the regioselectivity of acetyl xylan esterase from Bacillus pumilus as a catalyst for the deacetylation of glycoside acetates[J]. | Moen A R, Anthonsen T. 2009. Biocatalysis and Biotransformation. 27(3): 226-236.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Methyl 2,3,6-Tri-O-acetyl-α-D-glucopyranoside, 25 mg | sc-218756 | 25 mg | $300.00 |
