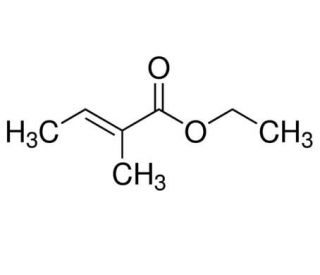
Ethyl tiglate (CAS 5837-78-5)
QUICK LINKS
Ethyl tiglate (ET) is an alkylating agent with a high reactivity that has found extensive use in scientific research and laboratory experiments. It is capable of introducing diverse chemical modifications to molecules, thus being an asset in studies involving DNA and protein alterations, gene expression, and cell signaling. Moreover, Ethyl tiglate has been utilized to investigate the effects of environmental toxins and various substances on cells. Notably, Ethyl tiglate can interact with nucleophilic sites on proteins and DNA, leading to the formation of covalent bonds. Such bonds can subsequently induce numerous changes in the structure and function of proteins and DNA, making Ethyl tiglate useful in understanding these critical biological components.
Ethyl tiglate (CAS 5837-78-5) References
- Metabolism of ethyl tiglate in apple fruits leads to the formation of small amounts of (R)-ethyl 2-methylbutanoate. | Hauck, T., et al. 2000. Enantiomer. 5: 505-12. PMID: 11143814
- Chemical composition, cytotoxicity effect and antimicrobial activity of Ceratonia siliqua essential oil with preservative effects against Listeria inoculated in minced beef meat. | Hsouna, AB., et al. 2011. Int J Food Microbiol. 148: 66-72. PMID: 21601302
- Isolation and identification of host cues from mango, Mangifera indica, that attract gravid female oriental fruit fly, Bactrocera dorsalis. | Jayanthi, PD., et al. 2012. J Chem Ecol. 38: 361-9. PMID: 22438014
- Aggregation pheromones inDrosophila borealis andDrosophila littoralis. | Bartelt, RJ., et al. 1988. J Chem Ecol. 14: 1319-27. PMID: 24276213
- Ester components of aggregation pheromone ofDrosophila virilis (Diptera: Drosophilidae). | Bartelt, RJ., et al. 1985. J Chem Ecol. 11: 1197-208. PMID: 24310382
- Specific volatile compounds from mango elicit oviposition in gravid Bactrocera dorsalis females. | Kamala Jayanthi, PD., et al. 2014. J Chem Ecol. 40: 259-66. PMID: 24623046
- Physiological and biochemical characteristics of the ethyl tiglate production pathway in the yeast Saprochaete suaveolens. | Grondin, E., et al. 2015. Yeast. 32: 57-66. PMID: 25407290
- A comparative study on the potential of epiphytic yeasts isolated from tropical fruits to produce flavoring compounds. | Grondin, E., et al. 2015. Int J Food Microbiol. 203: 101-8. PMID: 25802220
- Pollination of Specklinia by nectar-feeding Drosophila: the first reported case of a deceptive syndrome employing aggregation pheromones in Orchidaceae. | Karremans, AP., et al. 2015. Ann Bot. 116: 437-55. PMID: 26071932
- Benzaldehyde Synergizes the Response of Female Xyleborinus saxesenii (Coleoptera: Curculionidae, Scolytinae) to Ethanol. | Yang, CY., et al. 2018. J Econ Entomol. 111: 1691-1695. PMID: 29767753
- Selection by UV Mutagenesis and Physiological Characterization of Mutant Strains of the Yeast Saprochaete suaveolens (Former Geotrichum fragrans) with Higher Capacity to Produce Flavor Compounds. | Tan, M., et al. 2021. J Fungi (Basel). 7: PMID: 34947014
- The oxygen initial dip in the brain of anesthetized and awake mice. | Aydin, AK., et al. 2022. Proc Natl Acad Sci U S A. 119: e2200205119. PMID: 35353622
- An odorant binding protein mediates Bactrocera dorsalis olfactory sensitivity to host plant volatiles and male attractant compounds. | Liu, Z., et al. 2022. Int J Biol Macromol. 219: 538-544. PMID: 35907466
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ethyl tiglate, 100 g | sc-471918 | 100 g | $76.00 |
