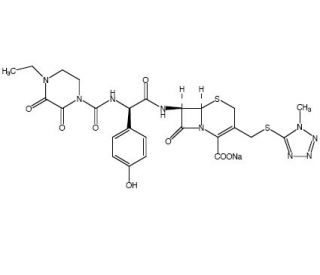
Cefoperazone sodium (CAS 62893-20-3)
See product citations (1)
QUICK LINKS
Cefoperazone Sodium, a semisynthetic, broad-spectrum, third-generation cephalosporin antibiotic, is noteworthy for its extensive application in microbiological research and bacterial culture studies. Unlike many other antibiotics, Cefoperazone Sodium is particularly resistant to beta-lactamase enzymes, making it effective against a wide range of Gram-positive and Gram-negative bacteria. Its mechanism of action involves inhibiting bacterial cell wall synthesis by binding to penicillin-binding proteins (PBPs), which are essential for maintaining the structural integrity of the bacterial cell wall. This inhibition leads to the weakening and eventual rupture of the cell wall, culminating in bacterial cell death. Cefoperazone Sodium is frequently used for selecting bacterial strains in genetic engineering and molecular biology experiments. Its resistance to beta-lactamase degradation enables researchers to employ it in environments where other beta-lactam antibiotics might fail. Additionally, its broad-spectrum activity is in studying polymicrobial cultures and in identifying and isolating bacteria that are resistant to other cephalosporins or penicillins.
Cefoperazone sodium (CAS 62893-20-3) References
- Chemiluminescence flow-injection analysis of beta-lactam antibiotics using the luminol-permanganate reaction. | Li, Y. and Lu, J. 2006. Luminescence. 21: 251-5. PMID: 16791833
- Cefoperazone sodium impregnated polycaprolactone composite implant for osteomyelitis. | Anand, A., et al. 2009. Indian J Pharm Sci. 71: 377-81. PMID: 20502542
- Liquid chromatography/tandem mass spectrometry assay for the simultaneous determination of cefoperazone and sulbactam in plasma and its application to a pharmacokinetic study. | Zhou, Y., et al. 2010. J Chromatogr B Analyt Technol Biomed Life Sci. 878: 3119-24. PMID: 20971044
- [Determination of target compounds in cefoperazone sodium and tazobactam sodium for injection by capillary electrophoresis]. | Jiang, R. and Sun, G. 2012. Se Pu. 30: 103-6. PMID: 22667102
- Cefoperazone sodium liposomal formulation to mitigate P. aeruginosa biofilm in Cystic fibrosis infection: A QbD approach. | Ghodake, V., et al. 2020. Int J Pharm. 587: 119696. PMID: 32736020
- Green and efficient degradation of cefoperazone sodium by Bi4O5Br2 leading to the production of non-toxic products: Performance and degradation pathway. | Chen, Y., et al. 2021. J Environ Sci (China). 100: 203-215. PMID: 33279033
- A case report of rash induced by cefoperazone sodium and sulbactam sodium plus metronidazole sodium chloride combined with morphine hydrochloride. | Huang, J., et al. 2020. Ann Palliat Med. 9: 4377-4383. PMID: 33302688
- Influence of Xuebijing injection combined with cefoperazone sodium and sulbactam sodium in treating hepatitis B-induced liver cirrhosis complicated by spontaneous bacterial peritonitis: TNF-α, IL-18, IL-6, and hepatic function. | Qian, D. and Zhang, Y. 2021. Am J Transl Res. 13: 2907-2914. PMID: 34017455
- Cefoperazone sodium/sulbactam sodium vs piperacillin sodium/tazobactam sodium for treatment of respiratory tract infection in elderly patients. | Wang, XX., et al. 2021. World J Clin Cases. 9: 8694-8701. PMID: 34734047
- Model-Informed Drug Development of New Cefoperazone Sodium and Sulbactam Sodium Combination (3:1): Pharmacokinetic/Pharmacodynamic Analysis and Antibacterial Efficacy Against Enterobacteriaceae. | Ji, XW., et al. 2022. Front Pharmacol. 13: 856792. PMID: 35924047
- Pharmacovigilance of cutaneous adverse drug reactions in associations with drugs and medical conditions: a retrospective study of hospitalized patients. | Zheng, L., et al. 2022. BMC Pharmacol Toxicol. 23: 62. PMID: 35948985
- Differential Proteomic and Genomic Comparison of Resistance Mechanism of Pseudomonas aeruginosa to Cefoperazone Sodium/Sulbactam Sodium. | Cai, S., et al. 2022. An Acad Bras Cienc. 94: e20211160. PMID: 36074406
- Compatibility of remifentanil hydrochloride with selected drugs during simulated Y-site administration. | Trissel, LA., et al. 1997. Am J Health Syst Pharm. 54: 2192-6. PMID: 9331439
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cefoperazone sodium, 1 g | sc-204678 | 1 g | $87.00 | |||
Cefoperazone sodium, 5 g | sc-204678A | 5 g | $250.00 |
