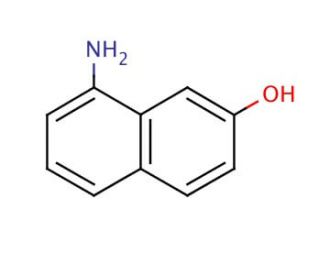
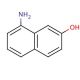
8-Amino-2-naphthol (CAS 118-46-7)
QUICK LINKS
8-Amino-2-naphthol is a chiral organocatalyst. 8-Amino-2-naphthol, an aromatic heterocyclic compound, exhibits solubility in polar organic solvents like dimethylformamide, dimethyl sulfoxide, and dimethylacetamide. This compound plays a role as an intermediate in the synthesis of diverse dyes and pigments. In scientific research, it has served as a substrate for studying enzymes involved in the metabolism of aromatic compounds, such as cytochrome P450. Furthermore, 8-Amino-2-naphthol has found applications as a reagent in the synthesis of other compounds, including indole derivatives and heterocyclic compounds. Notably, its utility extends to serving as a fluorescent probe for the detection of proteins, nucleic acids, and other biomolecules.
8-Amino-2-naphthol (CAS 118-46-7) References
- SYNTHETIC SCHISTOSOMICIDES. III. 5-(4-AMINO-1-NAPHTHYLAZO)URACIL AND RELATED HETEROCYCLIC AZO COMPOUNDS. | ELSLAGER, EF. and WORTH, DF. 1963. J Med Chem. 6: 444-7. PMID: 14184903
- Chirally aminated 2-naphthols--organocatalytic synthesis of non-biaryl atropisomers by asymmetric Friedel-Crafts amination. | Brandes, S., et al. 2006. Angew Chem Int Ed Engl. 45: 1147-51. PMID: 16389601
- Non-biaryl atropisomers in organocatalysis. | Brandes, S., et al. 2006. Chemistry. 12: 6039-52. PMID: 16789058
- Enantioselective synthesis of chiral tripodal cage compounds by [2 + 2 + 2] cycloaddition of branched triynes. | Shibata, T., et al. 2009. Org Lett. 11: 3906-8. PMID: 19663465
- 5-Amino-1-naphthol: two-dimensional sheets built up from R(4)(4)(18) rings formed by O-H..N, N-H..O and pi-pi interactions. | Rozycka-Sokolowska, E. and Marciniak, B. 2009. Acta Crystallogr C. 65: o565-8. PMID: 19893237
- Photochemistry and excited state prototropic behaviour of 8-amino 2-naphthol. | Gahlaut, R., et al. 2013. Spectrochim Acta A Mol Biomol Spectrosc. 109: 164-72. PMID: 23523759
- Photochemical Reactions of Aminonaphthols Caused by Laser Desorption/Ionization. | Nagoshi, K., et al. 2016. Mass Spectrom (Tokyo). 5: A0048. PMID: 27563510
- pH switch for OH-photoacidity in 5-amino-2-naphthol and 8-amino-2-naphthol. | Groves, MS., et al. 2018. Phys Chem Chem Phys. 20: 21325-21333. PMID: 30088501
- Divergent Hammett Plots of the Ground- and Excited-State Proton Transfer Reactions of 7-Substituted-2-Naphthol Compounds. | Cotter, LF., et al. 2019. J Phys Chem B. 123: 4301-4310. PMID: 31021637
- Chemical oxidative polymerization, optical, electrochemical and kinetic studies of 8-amino-2-naphthol. | Doğan, F., Kaya, İ., Bilici, A., & Yıldırım, M. 2015. Journal of Polymer Research. 22: 1-10.
- Synthesis, characterisation, solvatochromic behaviour and thermal decomposition kinetics of novel polyazo dyes containing amide group and their transition metal complexes | Çanakçı, D. 2019. Journal of Molecular Structure. 1181: 493-506.
- Ultralight self-charging triboelectric power paper with enhanced on-chip energy storage | Ma, W., Zhang, M., Yan, W., Zhu, J., Liu, J., & Song, W. 2022. Nano Energy. 101: 107601.
- Synthesis, characterization, photophysical and surface properties of poly(amino naphthalene) and its poly(azomethine) compound | Kaya, İ., Akyüz, B., & Özel, Ş. 2023. Journal of Applied Polymer Science. 140(27): e54021.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
8-Amino-2-naphthol, 25 g | sc-257014 | 25 g | $86.00 |
