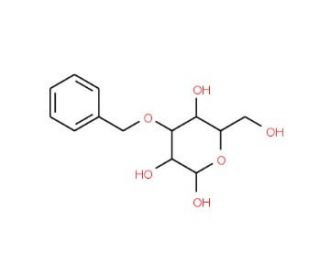
3-O-Benzyl-D-glucopyranose (CAS 10230-17-8)
QUICK LINKS
3-O-Benzyl-D-glucopyranose is extensively utilized in synthetic chemistry, particularly in the preparation of glycoconjugates and the study of carbohydrate chemistry. This chemical serves as an essential building block for the synthesis of more complex glycosidic structures, enabling researchers to modify and stabilize carbohydrate moieties for in-depth studies. The benzyl group at the 3-position specifically provides a protective function, preventing unwanted reactions at this site while allowing selective modifications at other positions of the glucose molecule. This specificity is crucial in the stepwise construction of oligosaccharides and glycoconjugates, which are fundamental to exploring biological recognition processes and cellular interactions. Moreover, 3-O-Benzyl-D-glucopyranose has been pivotal in developing new methodologies for glycosylation reactions, offering insights into enzyme mimicry and catalysis in organic synthesis. The use of this compound in research has significantly advanced the understanding of glycosidic bond formation and the role of carbohydrates in complex synthetic systems, contributing to the broader field of organic and carbohydrate chemistry. This focus on fundamental research helps to explain the underlying mechanisms governing chemical synthesis and molecular interaction.
3-O-Benzyl-D-glucopyranose (CAS 10230-17-8) References
- Synthesis of di- to hexasaccharide 1,2-linked beta-mannopyranan oligomers, a terminal S-linked tetrasaccharide congener and the corresponding BSA glycoconjugates. | Nitz, M. and Bundle, DR. 2001. J Org Chem. 66: 8411-23. PMID: 11735519
- An improved method for the preparation of 3-O-benzyl-6-O-pivaloyl-alpha-D-glucopyranose 1,2,4-orthopivalate. | Karakawa, M. and Nakatsubo, F. 2002. Carbohydr Res. 337: 951-4. PMID: 12007478
- Synthesis of (13)C-perlabeled cellulose with more than 99% isotopic enrichment by a cationic ring-opening polymerization approach. | Adelwöhrer, C., et al. 2009. Biomacromolecules. 10: 2817-22. PMID: 19754135
- A carbohydrate-based approach for the total synthesis of (-)-dinemasone B, (+)-4a-epi-dinemasone B, (-)-7-epi-dinemasone B, and (+)-4a,7-Di-epi-dinemasone B. | Xue, X., et al. 2013. J Org Chem. 78: 9354-65. PMID: 23987228
- Diastereoselective hydroboration of substituted exo-glucals revisited. A convenient route for the preparation of L-iduronic acid derivatives. | Rochepeau-Jobron, Laurence, and Jean-Claude Jacquinet. 1997. Carbohydrate research. 303.4: 395-406.
- Synthesis of 2-deoxy-α-DAH based on diazo chemistry by insertion reactions of 2-diazo-3-deoxy-d-arabino-heptulosonate derivatives mediated by rhodium (II). | Sarabia, F., S. Chammaa, and FJ López Herrera. 2001. Tetrahedron. 57.52: 10271-10279.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
3-O-Benzyl-D-glucopyranose, 500 mg | sc-283830 | 500 mg | $150.00 | |||
3-O-Benzyl-D-glucopyranose, 1 g | sc-283830A | 1 g | $300.00 |
