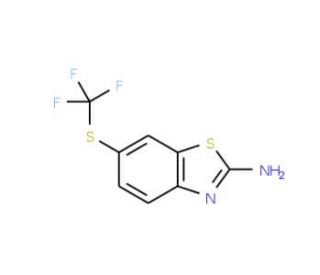
2-Amino-6-(trifluoromethylthio)benzothiazole (CAS 326-45-4)
QUICK LINKS
2-Amino-6-(trifluoromethylthio)benzothiazole is extensively used in chemical research, particularly in the synthesis of heterocyclic compounds. Its role as a building block in organic synthesis allows for the creation of a variety of benzothiazole derivatives that are studied for their chemical properties and reactivities. This compound is also pivotal in materials science, where it contributes to the development of novel materials with potential applications in electronics and photonics. In addition, 2-Amino-6-(trifluoromethylthio)benzothiazole is employed in the study of fluorescence properties, providing insights into molecular interactions and the design of fluorescent sensors. Its use in catalysis research is also notable, particularly in exploring its potential as a catalyst or a catalyst support for various chemical reactions, aiding in the development of more efficient and sustainable chemical processes.
2-Amino-6-(trifluoromethylthio)benzothiazole (CAS 326-45-4) References
- Naphtho[1,2-d]thiazol-2-ylamine (SKA-31), a new activator of KCa2 and KCa3.1 potassium channels, potentiates the endothelium-derived hyperpolarizing factor response and lowers blood pressure. | Sankaranarayanan, A., et al. 2009. Mol Pharmacol. 75: 281-95. PMID: 18955585
- The riluzole derivative 2-amino-6-trifluoromethylthio-benzothiazole (SKA-19), a mixed KCa2 activator and NaV blocker, is a potent novel anticonvulsant. | Coleman, N., et al. 2015. Neurotherapeutics. 12: 234-49. PMID: 25256961
- Rapid throughput analysis demonstrates that chemicals with distinct seizurogenic mechanisms differentially alter Ca2+ dynamics in networks formed by hippocampal neurons in culture. | Cao, Z., et al. 2015. Mol Pharmacol. 87: 595-605. PMID: 25583085
- SK channel inhibition mediates the initiation and amplitude modulation of synchronized burst firing in the spinal cord. | Mahrous, AA. and Elbasiouny, SM. 2017. J Neurophysiol. 118: 161-175. PMID: 28356481
- Is Targeting of Compensatory Ion Channel Gene Expression a Viable Therapeutic Strategy for Dravet Syndrome? | Isom, LL. 2019. Epilepsy Curr. 19: 193-195. PMID: 31035820
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Amino-6-(trifluoromethylthio)benzothiazole, 250 mg | sc-306635 | 250 mg | $170.00 | |||
2-Amino-6-(trifluoromethylthio)benzothiazole, 1 g | sc-306635A | 1 g | $514.00 |
