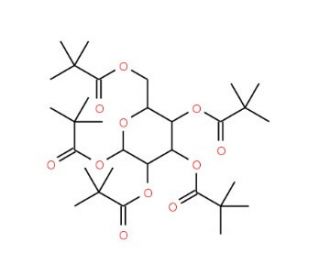
1,2,3,4,6-Penta-O-pivaloyl-D-mannopyranose (CAS 220017-47-0)
QUICK LINKS
1,2,3,4,6-Penta-O-pivaloyl-D-mannopyranose is a per-acylated derivative of mannose frequently utilized in carbohydrate chemistry. The pivaloyl groups serve as bulky protecting groups that shield the hydroxyl functionalities of D-mannopyranose, stabilizing the molecule for targeted glycosylation reactions. This compound provides a stable framework for synthesizing complex carbohydrate structures by allowing controlled deprotection and selective glycosylation. Researchers leverage it to study enzyme-catalyzed processes, focusing on the regioselectivity of glycosyltransferases and glycosidases. In research, 1,2,3,4,6-Penta-O-pivaloyl-D-mannopyranose is valuable for synthesizing glycomimetics and oligosaccharides. Its pivaloyl groups enhance solubility in nonpolar solvents, promoting compatibility with hydrophobic glycosyl acceptors during chemical synthesis. This enables researchers to construct carbohydrate structures relevant to investigating protein-carbohydrate interactions involved in cellular adhesion and signal transduction. Additionally, this derivative can help explain the enzymatic mechanisms of glycosidases by acting as a model substrate, providing insights into substrate specificity and catalytic efficiency. Overall, this compound′s versatility in carbohydrate synthesis makes it essential for exploring the roles of mannose-containing glycans in cellular and molecular processes, helping scientists advance their understanding of glycan-related biological pathways and signaling mechanisms.
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
1,2,3,4,6-Penta-O-pivaloyl-D-mannopyranose, 1 g | sc-287229 | 1 g | $374.00 |
