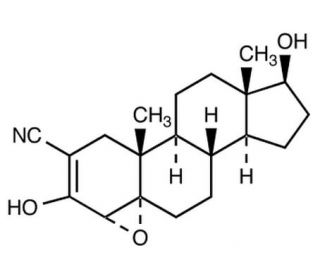
Molecular structure of Trilostane, CAS Number: 13647-35-3
Trilostane (CAS 13647-35-3)
See product citations (2)
Application:
Trilostane is A 3β-hydroxysteroid dehydrogenase inhibitor
CAS Number:
13647-35-3
Purity:
≥99%
Molecular Weight:
329.43
Molecular Formula:
C20H27NO3
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Product Citations
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
Trilostane is an inhibitor of isoforms 3β-HSD and 3β-HSD2 of the enzyme 3β-hydroxysteroid dehydrogenase, which plays a role in adrenal steroid biosynthesis. Investigators believe that Arg195 in 3β-HSD versus Pro195 in 3β-HSD2 attributes to the competitive inhibition of 3β-HSD, but not 3β-HSD2 by Trilostane. Research shows that trilostane is effective in suppressing the intracellular production of androstenedione, testosterone, and dihydrotestosterone in LNCaP-FGC prostate cancer cells and inhibiting cortisol biosynthesis in the human adrenal gland. This product serves as a useful tool in steroid biosynthesis research.
Trilostane (CAS 13647-35-3) References
- Differentiation-dependent progesterone synthesis and metabolism in NT2-N human neurons. | Pistritto, G., et al. 2009. Exp Neurol. 217: 302-11. PMID: 19306873
- 3beta-hydroxysteroid dehydrogenase is a possible pharmacological target in the treatment of castration-resistant prostate cancer. | Evaul, K., et al. 2010. Endocrinology. 151: 3514-20. PMID: 20534728
- Selective inhibition of human 3β-hydroxysteroid dehydrogenase type 1 as a potential treatment for breast cancer. | Thomas, JL., et al. 2011. J Steroid Biochem Mol Biol. 125: 57-65. PMID: 20736065
- Trilostane, an inhibitor of 3β-hydroxysteroid dehydrogenase, has an agonistic activity on androgen receptor in human prostate cancer cells. | Takizawa, I., et al. 2010. Cancer Lett. 297: 226-30. PMID: 20831980
- Treatment of Cushing's syndrome with trilostane (WIN 24,540), an inhibitor of adrenal steroid biosynthesis. | Komanicky, P., et al. 1978. J Clin Endocrinol Metab. 47: 1042-51. PMID: 233687
- Possible therapeutic effect of trilostane in rodent models of inflammation and nociception. | Tung, D., et al. 2013. Curr Ther Res Clin Exp. 75: 71-6. PMID: 24465047
- Long-term survival of dogs with adrenal-dependent hyperadrenocorticism: a comparison between mitotane and twice daily trilostane treatment. | Arenas, C., et al. 2014. J Vet Intern Med. 28: 473-80. PMID: 24495125
- Expression of 11β-hydroxysteroid dehydrogenase isoforms in canine adrenal glands treated with trilostane. | Teshima, T., et al. 2014. Vet J. 200: 452-5. PMID: 24745768
- Comparison of adrenocorticotropic hormone stimulation test results started 2 versus 4 hours after trilostane administration in dogs with naturally occurring hyperadrenocorticism. | Bonadio, CM., et al. 2014. J Vet Intern Med. 28: 1239-43. PMID: 24863172
- Prognostic factors for survival in dogs with pituitary-dependent hypercortisolism treated with trilostane. | Fracassi, F., et al. 2015. Vet Rec. 176: 49. PMID: 25170036
- Cortisol Concentrations in Well-Regulated Dogs with Hyperadrenocorticism Treated with Trilostane. | Midence, JN., et al. 2015. J Vet Intern Med. 29: 1529-33. PMID: 26374943
- Multicenter study of trilostane: a new hormonal agent in advanced postmenopausal breast cancer. | Williams, CJ., et al. 1987. Cancer Treat Rep. 71: 1197-201. PMID: 3690530
- Studies on the metabolism of trilostane, an inhibitor of adrenal steroidogenesis. | Mori, Y., et al. 1981. Chem Pharm Bull (Tokyo). 29: 2646-52. PMID: 7349283
Inhibitor of:
17β-HSD, 17β-HSD14, 17β-HSD2, 17β-HSD3, 17β-HSD6, 17β-HSD7, 3β-HSD, 3β-HSD2, 3β-HSD3, 3β-HSD4, 3β-HSD5, 3β-HSD7, 3beta-HSD, 4930458L03Rik, AKR1C13, Choriogonadotropin, CSH1, CYP11A1, CYP11B, CYP11B1, Cyp21a1, DEXI, EG384585, EG434676, EG545948, EG626305, EG667693, Estradiol, Etd, Fdx1, GCDFP-15, GLCCI1, Gonadotropin α, LOC100039517, MC2-R, MRAP, PLP-J, Purγ, SPATA32, StAR, StARD5, and ZP1.Activator of:
3β-HSD4, GMEB-1, and PLP-K.Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Trilostane, 10 mg | sc-208469 | 10 mg | $228.00 | |||
Trilostane, 100 mg | sc-208469A | 100 mg | $1217.00 |
