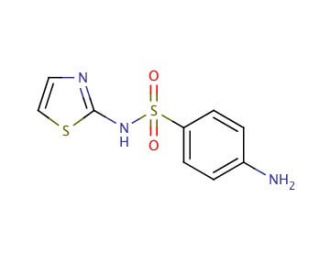
Sulfathiazole (CAS 72-14-0)
QUICK LINKS
Sulfathiazole is a compound that functions as a competitive inhibitor of dihydropteroate synthase, an enzyme involved in the folate synthesis pathway in bacteria. By inhibiting this enzyme, sulfathiazole disrupts the production of tetrahydrofolic acid, which is for the synthesis of nucleic acids and proteins in bacterial cells. This disruption ultimately leads to the inhibition of bacterial growth and reproduction. Sulfathiazole achieves this by binding to the active site of dihydropteroate synthase, preventing the enzyme from carrying out its normal function. As a result, the bacteria are unable to produce components for their survival and are ultimately unable to proliferate. This mechanism of action makes sulfathiazole useful for studying bacterial folate metabolism and for investigating potential targets for antibacterial agents.
Sulfathiazole (CAS 72-14-0) References
- Antibiotic immunosensing: determination of sulfathiazole in water and honey. | Jornet, D., et al. 2010. Talanta. 81: 1585-92. PMID: 20441943
- Fluorescence interaction and determination of sulfathiazole with trypsin. | Gökoğlu, E. and Yılmaz, E. 2014. J Fluoresc. 24: 1439-45. PMID: 25106475
- Sulfathiazole: analytical methods for quantification in seawater and macroalgae. | Leston, S., et al. 2015. Environ Toxicol Pharmacol. 39: 77-84. PMID: 25473819
- Evaluation of sulfathiazole degradation by persulfate in Milli-Q water and in effluent of a sewage treatment plant. | Velosa, AC. and Nascimento, CAO. 2017. Environ Sci Pollut Res Int. 24: 6270-6277. PMID: 27287494
- Sorption of sulfathiazole in the soil treated with giant Miscanthus-derived biochar: effect of biochar pyrolysis temperature, soil pH, and aging period. | Kim, H., et al. 2018. Environ Sci Pollut Res Int. 25: 25681-25689. PMID: 28455564
- Photodegradation of tetracycline and sulfathiazole individually and in mixtures. | Yun, SH., et al. 2018. Food Chem Toxicol. 116: 108-113. PMID: 29630946
- Removal of Sulfamethoxazole, Sulfathiazole and Sulfamethazine in their Mixed Solution by UV/H2O2 Process. | Zhu, G., et al. 2019. Int J Environ Res Public Health. 16: PMID: 31117187
- Waste-wood-derived biochar cathode and its application in electro-Fenton for sulfathiazole treatment at alkaline pH with pyrophosphate electrolyte. | Deng, F., et al. 2019. J Hazard Mater. 377: 249-258. PMID: 31170573
- Sulfate radical-based oxidation of the antibiotics sulfamethoxazole, sulfisoxazole, sulfathiazole, and sulfamethizole: The role of five-membered heterocyclic rings. | Zhou, L., et al. 2019. Sci Total Environ. 692: 201-208. PMID: 31344571
- Nitrogen-doped porous carbon encapsulating iron nanoparticles for enhanced sulfathiazole removal via peroxymonosulfate activation. | Chen, L., et al. 2020. Chemosphere. 250: 126300. PMID: 32113094
- Remediation of sulfathiazole contaminated soil by peroxymonosulfate: Performance, mechanism and phytotoxicity. | Zhang, Y., et al. 2022. Sci Total Environ. 830: 154839. PMID: 35341832
- Generation of high-valent iron-oxo porphyrin cation radicals on hemin loaded carbon nanotubes for efficient degradation of sulfathiazole. | Chen, S., et al. 2023. J Hazard Mater. 444: 130402. PMID: 36403452
- An FeP/carbon composite derived from a phytic acid-Fe3+ complex for sulfathiazole degradation through peroxymonosulfate activation. | Wang, X., et al. 2023. Chemosphere. 313: 137562. PMID: 36526142
- Physiological model for distribution of sulfathiazole in swine. | Duddy, J., et al. 1984. J Pharm Sci. 73: 1525-8. PMID: 6520750
- Dissolution studies of povidone-sulfathiazole coacervated systems. | Badawi, AA. and El-Sayed, AA. 1980. J Pharm Sci. 69: 492-7. PMID: 7381728
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Sulfathiazole, 100 mg | sc-215927 | 100 mg | $102.00 | |||
Sulfathiazole, 1 g | sc-215927A | 1 g | $82.00 |
