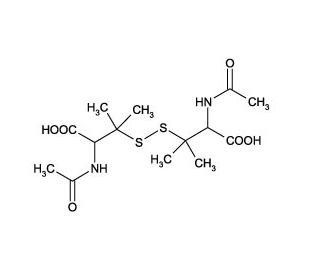
N-Acetyl-D,L-penicillamine disulfide
See product citations (1)
QUICK LINKS
N-Acetyl-D,L-penicillamine disulfide is a compound that functions as a chelating agent.It acts by forming stable complexes with metal ions. This interaction helps to remove excess metal ions from biological systems, making it useful for studying metal ion homeostasis and toxicity. The compound′s mechanism of action involves the formation of covalent bonds with metal ions, leading to the formation of stable complexes that can be analyzed and studied in various experiments. N-Acetyl-D,L-penicillamine disulfide has been used to investigate the role of metal ions in biological processes, providing insights into the molecular mechanisms underlying metal ion regulation and signaling pathways.
N-Acetyl-D,L-penicillamine disulfide References
- In vitro cytotoxicity of the nitric oxide donor, S-nitroso-N-acetyl-penicillamine, towards cells from human oral tissue. | Babich, H., et al. 1999. Pharmacol Toxicol. 84: 218-25. PMID: 10361978
- Nitric oxide affects the production of reactive oxygen species in hepatoma cells: implications for the process of oxygen sensing. | Genius, J. and Fandrey, J. 2000. Free Radic Biol Med. 29: 515-21. PMID: 11025195
- Different capacities of carp leukocytes to encounter nitric oxide-mediated stress: a role for the intracellular reduced glutathione pool. | Saeij, JP., et al. 2003. Dev Comp Immunol. 27: 555-68. PMID: 12697312
- The role of endogenous heme synthesis and degradation domain cysteines in cellular iron-dependent degradation of IRP2. | Bourdon, E., et al. 2003. Blood Cells Mol Dis. 31: 247-55. PMID: 12972033
- Nitric oxide inhibits the rate and strength of cardiac contractions in the lobster Homarus americanus by acting on the cardiac ganglion. | Mahadevan, A., et al. 2004. J Neurosci. 24: 2813-24. PMID: 15028775
- Contrasting potential of nitric oxide and peroxynitrite to mediate oligodendrocyte injury in multiple sclerosis. | Jack, C., et al. 2007. Glia. 55: 926-34. PMID: 17437305
- Nitric oxide donor s-nitroso-n-acetyl penicillamine (SNAP) alters meiotic spindle morphogenesis in Xenopus oocytes. | Gelaude, A., et al. 2015. J Cell Biochem. 116: 2445-54. PMID: 25923058
- Reduction of Thrombosis and Bacterial Infection via Controlled Nitric Oxide (NO) Release from S-Nitroso-N-acetylpenicillamine (SNAP) Impregnated CarboSil Intravascular Catheters. | Wo, Y., et al. 2017. ACS Biomater Sci Eng. 3: 349-359. PMID: 28317023
- Don't just say no: Differential pathways and pharmacological responses to diverse nitric oxide donors. | Chiesa, JJ., et al. 2018. Biochem Pharmacol. 156: 1-9. PMID: 30080991
- The nitric oxide--cyclic GMP pathway and synaptic depression in rat hippocampal slices. | Boulton, CL., et al. 1994. Eur J Neurosci. 6: 1528-35. PMID: 7850017
- Neurotoxicity in conscious rats following intraventricular SNAP, a nitric oxide donor. | Gross, PM., et al. 1994. Neuropharmacology. 33: 915-27. PMID: 7969812
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
N-Acetyl-D,L-penicillamine disulfide, 1 mg | sc-205957 | 1 mg | $37.00 | |||
N-Acetyl-D,L-penicillamine disulfide, 5 mg | sc-205957A | 5 mg | $93.00 |
