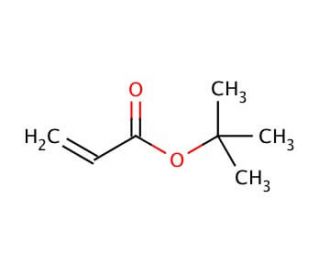
tert-Butyl acrylate (CAS 1663-39-4)
QUICK LINKS
Tert-Butyl Acrylate (TBA) is a colorless liquid with a sweet odor. This organic compound serves as a monomer in the production of plastics, coatings, and adhesives, while also finding use as an industrial solvent in applications like fuel additives and metalworking fluids. Additionally, tert-Butyl acrylate plays a role in synthesizing various compounds, including polymers and surfactants. Due to its versatile nature, tert-Butyl acrylate has become a subject of extensive study in numerous scientific fields. It has been employed as a model compound for exploring the kinetics and thermodynamics of polymerization reactions. Furthermore, researchers have utilized it to investigate how solvents influence polymer properties and how copolymerization impacts polymer characteristics. Notably, tert-Butyl acrylate plays a significant role in the synthesis of various polymers, such as polyurethanes, polyamides, and polyesters. The polymerization reaction of tert-Butyl acrylate is initiated by adding a catalyst like sulfuric acid, resulting in an exothermic process. Usually carried out at temperatures of 150°C or higher, the reaction rate is accelerated by raising the temperature. As the reaction progresses, monomer molecules join the growing polymer chain, leading to the formation of the desired polymers.
tert-Butyl acrylate (CAS 1663-39-4) References
- Hydrogen Bonding-Derived Healable Polyacrylate Elastomers via On-demand Copolymerization of n-Butyl Acrylate and tert-Butyl Acrylate. | Wang, W., et al. 2020. ACS Appl Mater Interfaces. 12: 50812-50822. PMID: 33118819
- Interaction of Proteins with a Planar Poly(acrylic acid) Brush: Analysis by Quartz Crystal Microbalance with Dissipation Monitoring (QCM-D). | Walkowiak, J., et al. 2020. Polymers (Basel). 13: PMID: 33396873
- Catalytic enantioselective reductive domino alkyl arylation of acrylates via nickel/photoredox catalysis. | Qian, P., et al. 2021. Nat Commun. 12: 6613. PMID: 34785647
- A Sprayable and Visible Light Rapid-Cured Strippable Film for Surface Radioactive Decontamination. | Zhang, H., et al. 2022. Polymers (Basel). 14: PMID: 35267831
- PdII -Catalyzed C(alkenyl)-H Activation Facilitated by a Transient Directing Group. | Liu, M., et al. 2022. Angew Chem Int Ed Engl. 61: e202203624. PMID: 35467792
- Emulsion-Templated Porous Polymers for Efficient Dye Removal. | Üzüm, G., et al. 2022. ACS Omega. 7: 16127-16140. PMID: 35571856
- Preparation and controlled-release properties of a dual-response acidizing corrosion inhibitor. | Zhitao, Y., et al. 2022. RSC Adv. 12: 27055-27063. PMID: 36320837
- Ethylene-Vinyl Acetate Copolymers as Potential Thermoplastic Modifiers of Photopolymer Compositions. | Bazhanov, DA., et al. 2022. Polymers (Basel). 15: PMID: 36616485
- Visible-Light-Controlled Histone Deacetylase Inhibitors for Targeted Cancer Therapy. | Josa-Culleré, L. and Llebaria, A. 2023. J Med Chem. 66: 1909-1927. PMID: 36654474
- PCL/Gelatin/Graphene Oxide Electrospun Nanofibers: Effect of Surface Functionalization on In Vitro and Antibacterial Response. | Hamdan, N., et al. 2023. Nanomaterials (Basel). 13: PMID: 36770449
- Ni-catalyzed arylation of alkynes with organoboronic acids and aldehydes to access stereodefined allylic alcohols. | Tao, SC., et al. 2023. Chem Sci. 14: 2040-2045. PMID: 36845934
- A co-anchoring strategy for the synthesis of polar bimodal polyethylene. | Zou, C., et al. 2023. Nat Commun. 14: 1442. PMID: 36922533
- Triarylamminium Radical Cation Facilitates the Deprotection of tert-Butyl Groups in Esters, Ethers, Carbonates, and Carbamates. | Hidasová, D. and Slanina, T. 2023. J Org Chem. 88: 6932-6938. PMID: 37126731
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
tert-Butyl acrylate, 25 ml | sc-253632 | 25 ml | $45.00 | |||
tert-Butyl acrylate, 100 ml | sc-253632A | 100 ml | $53.00 |
