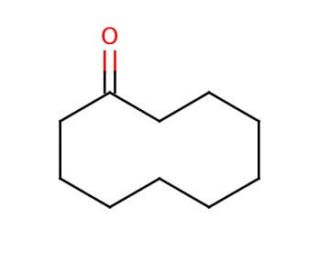
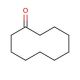
Cyclodecanone (CAS 1502-06-3)
QUICK LINKS
Cyclodecanone is a cyclic ketone that is of particular interest to researchers engaged in the study of macrocyclic and supramolecular chemistry. It acts as a building block for the synthesis of a wide range of macrocyclic compounds, which have applications in materials science and host-guest chemistry. Investigations involving cyclodecanone often focus on its ring-opening polymerization, which is key to understanding the behavior of cyclic monomers in the formation of polymers with unique properties. The compound′s reactivity is also studied in the context of organic synthesis, where it is used to produce larger, complex structures through various chemical reactions. Additionally, cyclodecanone is employed in the exploration of fragrance chemistry, due to its potential as an intermediate in the synthesis of musk odorants and other aroma compounds.
Cyclodecanone (CAS 1502-06-3) References
- Reaction of arylsulfonylhydrazones of aldehydes with alpha-magnesio sulfones. A novel olefin synthesis. | Kurek-Tyrlik, A., et al. 2001. J Org Chem. 66: 6994-7001. PMID: 11597219
- Thujone--cause of absinthism? | Lachenmeier, DW., et al. 2006. Forensic Sci Int. 158: 1-8. PMID: 15896935
- Beyond the corey reaction: one-step diolefination of cyclic ketones. | Butova, ED., et al. 2007. J Org Chem. 72: 5689-96. PMID: 17580903
- Enantioselective total synthesis and confirmation of the absolute and relative stereochemistry of streptorubin B. | Hu, DX., et al. 2011. J Am Chem Soc. 133: 1799-804. PMID: 21166419
- Determination of the biologically active flavour substances thujone and camphor in foods and medicines containing sage (Salvia officinalis L.). | Walch, SG., et al. 2011. Chem Cent J. 5: 44. PMID: 21777420
- Reactions of OH radicals with C6-C10 cycloalkanes in the presence of NO: isomerization of C7-C10 cycloalkoxy radicals. | Aschmann, SM., et al. 2011. J Phys Chem A. 115: 14452-61. PMID: 22128790
- Mammalian odorant receptor tuning breadth persists across distinct odorant panels. | Kepchia, D., et al. 2017. PLoS One. 12: e0185329. PMID: 28945824
- Total Synthesis of Marine Alkaloids Motuporamines A and B via Ring Expansion of Cyclic β-Keto Esters. | Song, ZJ., et al. 2021. ACS Omega. 6: 881-888. PMID: 33458539
- Analytical Separation of Carcinogenic and Genotoxic Alkenylbenzenes in Foods and Related Products (2010-2020). | Dang, HNP. and Quirino, JP. 2021. Toxins (Basel). 13: PMID: 34071244
- Ozonation of Decalin as a Model Saturated Cyclic Molecule: A Spectroscopic Study. | Ershov, BG., et al. 2021. Molecules. 26: PMID: 34577038
- New Strategy for Inducing Resistance against Bacterial Wilt Disease Using an Avirulent Strain of Ralstonia solanacearum. | Moussa, Z., et al. 2022. Microorganisms. 10: PMID: 36144416
- Receptor cell responses to odorants: similarities and differences among odorants. | Sicard, G. and Holley, A. 1984. Brain Res. 292: 283-96. PMID: 6692160
- Enhancement of cyclodextrin production through use of debranching enzymes. | Rendleman, JA. 1997. Biotechnol Appl Biochem. 26: 51-61. PMID: 9262003
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cyclodecanone, 1 g | sc-239604 | 1 g | $510.00 |
