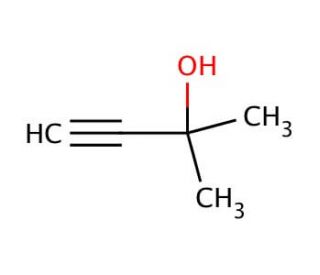
2-Methyl-3-butyn-2-ol (CAS 115-19-5)
QUICK LINKS
2-Methyl-3-butyn-2-ol, a synthetic organic compound characterized by its unique alkyne and alcohol functional groups, plays a significant role in various research domains, particularly in the synthesis of complex organic molecules. Its structure allows for versatile chemical reactions, making it a valuable building block in organic chemistry. Researchers often utilize 2-Methyl-3-butyn-2-ol for its ability to undergo reactions such as hydrolysis and oxidation, which are for modifying molecular frameworks and introducing functional groups during synthetic pathways. Moreover, its presence in the creation of polymers, fragrances, and specialty chemicals underscores its importance. Its utility in research extends to catalysis studies, where it serves as a substrate for testing the efficacy and selectivity of novel catalysts. This compound exemplifies the foundational tools in chemical research, facilitating advancements in material science and synthetic organic chemistry through its broad application in constructing complex chemical entities and exploring reaction mechanisms.
2-Methyl-3-butyn-2-ol (CAS 115-19-5) References
- Enantioselective addition of 2-methyl-3-butyn-2-ol to aldehydes: preparation of 3-hydroxy-1-butynes. | Boyall, D., et al. 2000. Org Lett. 2: 4233-6. PMID: 11150207
- Regioselective synthesis of 6-alkyl- and 6-prenylpolyhydroxyisoflavones and 6-alkylcoumaronochromone derivatives. | Tsukayama, M., et al. 2004. Chem Pharm Bull (Tokyo). 52: 1285-9. PMID: 15516747
- Ligand accelerated indium(III)-catalyzed asymmetric alkynylation of aldehydes with 2-methyl-3-butyn-2-ol as an ethyne equivalent donor. | Harada, S., et al. 2007. Chem Commun (Camb). 948-50. PMID: 17311130
- Detection of estrus in Indian blackbuck: behavioural, hormonal and urinary volatiles evaluation. | Archunan, G. and Rajagopal, T. 2013. Gen Comp Endocrinol. 181: 156-66. PMID: 23229002
- Influence of natural adsorbates of magnesium oxide on its reactivity in basic catalysis. | Cornu, D., et al. 2013. Phys Chem Chem Phys. 15: 19870-8. PMID: 24145744
- Practical synthesis of aryl-2-methyl-3-butyn-2-ols from aryl bromides via conventional and decarboxylative copper-free Sonogashira coupling reactions. | Caporale, A., et al. 2014. Beilstein J Org Chem. 10: 384-93. PMID: 24605159
- Ultrasound-assisted selective hydrogenation of C-5 acetylene alcohols with Lindlar catalysts. | Tripathi, B., et al. 2015. Ultrason Sonochem. 26: 445-451. PMID: 25797157
- Asymmetric 1,4-bis(ethynyl)bicyclo[2.2.2]octane rotators via monocarbinol functionalization. Ready access to polyrotors. | Lemouchi, C. and Batail, P. 2015. Beilstein J Org Chem. 11: 1881-5. PMID: 26664606
- A screen for kinase inhibitors identifies antimicrobial imidazopyridine aminofurazans as specific inhibitors of the Listeria monocytogenes PASTA kinase PrkA. | Schaenzer, AJ., et al. 2017. J Biol Chem. 292: 17037-17045. PMID: 28821610
- Enantioselective Catalytic Asymmetric A3 Coupling with Phosphino-Imidazoline Ligands. | Rokade, BV. and Guiry, PJ. 2019. J Org Chem. 84: 5763-5772. PMID: 30942080
- Observation of Substituent Effects in the Electrochemical Adsorption and Hydrogenation of Alkynes on Pt{hkl} Using SHINERS. | Guan, S., et al. 2020. ACS Catal. 10: 10999-11010. PMID: 33042608
- Dynamic Modification of Palladium Catalysts with Chain Alkylamines for the Selective Hydrogenation of Alkynes. | Luo, Q., et al. 2021. ACS Appl Mater Interfaces. 13: 31775-31784. PMID: 34227385
- Understanding of the interactions between azole-anion-based ionic liquids and 2-methyl-3-butyn-2-ol from the experimental perspective: the cage effect. | Fu, X., et al. 2022. Phys Chem Chem Phys. 24: 12550-12562. PMID: 35579063
- Breaking scaling relationships in alkynol semi-hydrogenation by manipulating interstitial atoms in Pd with d-electron gain. | Yang, Y., et al. 2022. Nat Commun. 13: 2754. PMID: 35585084
- 2-Methyl-3-butyn-2-ol as an acetylene precursor in the Mannich reaction. A new synthesis of suicide inactivators of monoamine oxidase. | Fowler, JS. 1977. J Org Chem. 42: 2637-7. PMID: 874623
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methyl-3-butyn-2-ol, 5 ml | sc-238135 | 5 ml | $27.00 | |||
2-Methyl-3-butyn-2-ol, 100 ml | sc-238135A | 100 ml | $20.00 |
