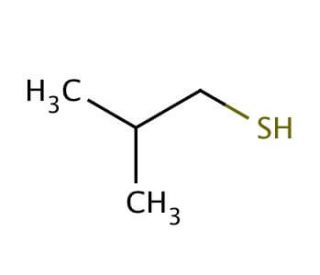
Molecular structure of 2-Methyl-1-propanethiol, CAS Number: 513-44-0
2-Methyl-1-propanethiol (CAS 513-44-0)
Alternate Names:
Isobutyl mercaptan
Application:
2-Methyl-1-propanethiol is used in the study of drug delivery and self-assembling monolayers
CAS Number:
513-44-0
Purity:
≥92%
Molecular Weight:
90.19
Molecular Formula:
(CH3)2CHCH2SH
Supplemental Information:
This is classified as a Dangerous Good for transport and may be subject to additional shipping charges.
For Research Use Only. Not Intended for Diagnostic or Therapeutic Use.
* Refer to Certificate of Analysis for lot specific data.
QUICK LINKS
Ordering Information
Description
Technical Information
Safety Information
SDS & Certificate of Analysis
2-methyl-1-propanethiol has various uses in research including, drug delivery, materials science, and nanoelectronics. It may act to attach with L-Cys (the carrier) via a disulfide bond to form the brain-targeted drug delivery systems (BTDS), and it is used in the study of self-assembling monolayers (SAMs). Moreover, its antioxidant properties make it valuable in neutralizing harmful free radicals. Additionally, 2-Methyl-1-propanethiol acts as a chelating agent, forming stable complexes with metal ions and influencing various biochemical processes.
2-Methyl-1-propanethiol (CAS 513-44-0) References
- Molar Refractivity and Connectivity Index Correlations for Henry's Law Virial Coefficients of Odorous Sulfur Compounds on Carbon and for Gas-Chromatographic Retention Indices. | Rybolt, TR., et al. 2001. J Colloid Interface Sci. 234: 168-177. PMID: 11161504
- Effect of pH and temperature on the kinetics of odor oxidation using chlorine dioxide. | Kastner, JR., et al. 2003. J Air Waste Manag Assoc. 53: 1218-24. PMID: 14604331
- On-column derivatization-capillary electrochromatography with o-phthalaldehyde/alkylthiol for assay of biogenic amines. | Oguri, S., et al. 2004. J Chromatogr A. 1044: 271-6. PMID: 15354448
- Transferable potentials for phase equilibria. 8. United-atom description for thiols, sulfides, disulfides, and thiophene. | Lubna, N., et al. 2005. J Phys Chem B. 109: 24100-7. PMID: 16375402
- Targeting the cerebrovascular large neutral amino acid transporter (LAT1) isoform using a novel disulfide-based brain drug delivery system. | Killian, DM., et al. 2007. Drug Deliv. 14: 25-31. PMID: 17107928
- Electrogenerated chemiluminescence from PbS quantum dots. | Sun, L., et al. 2009. Nano Lett. 9: 789-93. PMID: 19115964
- Synthesis of monodisperse [Oct4N(+)][Au25(SR)18(-)] nanoparticles, with some mechanistic observations. | Parker, JF., et al. 2010. Langmuir. 26: 13650-4. PMID: 20695616
- Chain-branching control of the atomic structure of alkanethiol-based gold-sulfur interfaces. | Wang, Y., et al. 2011. J Am Chem Soc. 133: 14856-9. PMID: 21859147
- Snake-venom resistance as a mammalian trophic adaptation: lessons from didelphid marsupials. | Voss, RS. and Jansa, SA. 2012. Biol Rev Camb Philos Soc. 87: 822-37. PMID: 22404916
- Unimolecular photochemistry of n-alkenes studied by photodissociation-photoionization mass spectrometry. | Van Bramer, SE., et al. 1993. J Am Soc Mass Spectrom. 4: 65-72. PMID: 24234746
- Smelling Sulfur: Copper and Silver Regulate the Response of Human Odorant Receptor OR2T11 to Low-Molecular-Weight Thiols. | Li, S., et al. 2016. J Am Chem Soc. 138: 13281-13288. PMID: 27659093
- Conformations and Low-Frequency Intramolecular Motions of 1-Butanol, 1-Butanethiol, Iso-butanol, and Iso-butanethiol Investigated by Fourier Transform Microwave Spectroscopy Combined with Quantum Chemical Calculations. | Kawashima, Y., et al. 2021. J Phys Chem A. 125: 1166-1183. PMID: 33502865
- Embedding alkenes within an icosahedral inorganic fullerene {(NH4)42[Mo132O372(L)30(H2O)72]} for trapping volatile organics. | Pow, RW., et al. 2020. Chem Sci. 11: 2388-2393. PMID: 34084401
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
2-Methyl-1-propanethiol, 100 ml | sc-230502 | 100 ml | $70.00 |
