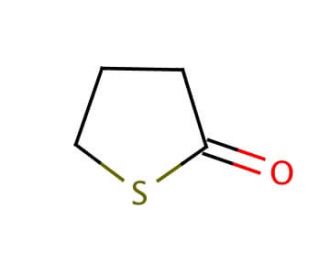
γ-Thiobutyrolactone (CAS 1003-10-7)
QUICK LINKS
γ-Thiobutyrolactone is a chemical compound that serves as a building block in organic synthesis and is used in various research applications. In synthetic chemistry, it acts as a precursor for the synthesis of γ-amino acids and their derivatives, which are of interest due to their presence in biologically active peptides. The reactivity of γ-thiobutyrolactone is exploited to create thiol-containing compounds, which are important in the study of molecular structure and function, as well as in the design of new materials and catalysts. Its lactone ring can undergo ring-opening reactions, allowing it to be incorporated into larger, more complex molecules. Researchers employ γ-thiobutyrolactone to investigate the mechanisms of lactone polymerization and the properties of the resulting polymers. Additionally, this compound is used in the development of new synthetic methodologies that may include the formation of carbon-sulfur bonds.
γ-Thiobutyrolactone (CAS 1003-10-7) References
- Catalytic synthesis of thiobutyrolactones via CO insertion into the C-S bond of thietanes in the presence of a heterodinuclear organoplatinum-cobalt complex. | Furuya, M., et al. 2003. Chem Commun (Camb). 2046-7. PMID: 12934904
- Modification of cellulose fiber surfaces by use of a lipase and a xyloglucan endotransglycosylase. | Gustavsson, MT., et al. 2005. Biomacromolecules. 6: 196-203. PMID: 15638521
- Mechanism of hydrolysis and aminolysis of homocysteine thiolactone. | Garel, J. and Tawfik, DS. 2006. Chemistry. 12: 4144-52. PMID: 16453352
- Relations of lysophosphatidylcholine in low-density lipoprotein with serum lipoprotein-associated phospholipase A2, paraoxonase and homocysteine thiolactonase activities in patients with type 2 diabetes mellitus. | Sonoki, K., et al. 2009. Diabetes Res Clin Pract. 86: 117-23. PMID: 19748147
- An improved synthesis of the selective EP4 receptor agonist ONO-4819. | Ohta, C., et al. 2009. J Org Chem. 74: 8298-308. PMID: 19803523
- The use of superporous Ac-CGGASIKVAVS-OH-modified PHEMA scaffolds to promote cell adhesion and the differentiation of human fetal neural precursors. | Kubinová, S., et al. 2010. Biomaterials. 31: 5966-75. PMID: 20483453
- Decreased paraoxonase 1 (PON1) lactonase activity in hemodialyzed and renal transplanted patients. A novel cardiovascular biomarker in end-stage renal disease. | Sztanek, F., et al. 2012. Nephrol Dial Transplant. 27: 2866-72. PMID: 22247228
- Thiolated hemicellulose as a versatile platform for one-pot click-type hydrogel synthesis. | Maleki, L., et al. 2015. Biomacromolecules. 16: 667-74. PMID: 25574855
- Circulating Fibroblast Growth Factor-23 Level and Paraoxonase-1 Lactonase Activity in Chronic Hemodialysis Patients: Their Impact on the Incidence of Native AV Fistula Thrombosis. | Zohny, SF., et al. 2016. Clin Invest Med. 39: E173-E181. PMID: 27805900
- Comparative analysis of the metal-dependent structural and functional properties of mouse and human SMP30. | Dutta, RK., et al. 2019. PLoS One. 14: e0218629. PMID: 31220150
- Lactone modulation of the gamma-aminobutyric acid A receptor: evidence for a positive modulatory site. | Williams, KL., et al. 1997. Mol Pharmacol. 52: 114-9. PMID: 9224820
- A Novel Synthesis of Poly(ester-alt-sulfide)s by the Ring-Opening Alternating Copolymerization of Oxiranes with gamma-Thiobutyrolactone Using Quaternary Onium Salts or Crown Ether Complexes as Catalysts. | Nishikubo, T., et al. 1998. Macromolecules. 31: 4746-52. PMID: 9680408
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
γ-Thiobutyrolactone, 10 g | sc-224308 | 10 g | $301.00 | |||
γ-Thiobutyrolactone, 50 g | sc-224308A | 50 g | $1173.00 |
