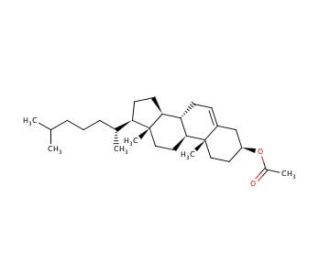
Cholesteryl acetate (CAS 604-35-3)
QUICK LINKS
Cholesteryl acetate (CA) stands as a versatile molecule derived from cholesterol, offering a wide array of applications and potential uses. As a lipid-soluble compound, it has found extensive utilization in research applications. Cholesteryl acetate exhibits a diverse range of biological activities and holds substantial potential for in vitro research. In vitro studies have utilized cholesteryl acetate to delve into the effects of cholesterol on cell cultures and to investigate its influence on diverse biochemical pathways. Despite the ongoing quest for a comprehensive understanding of its mechanism of action, it is known that cholesteryl acetate interacts with several proteins and enzymes within the body. Notably, it has been observed to inhibit the activity of key enzymes such as HMG-CoA reductase and fatty acid synthase, which play pivotal roles in cholesterol synthesis.
Cholesteryl acetate (CAS 604-35-3) References
- In vitro fibrillogenesis of the amyloid beta 1-42 peptide: cholesterol potentiation and aspirin inhibition. | Harris, JR. 2002. Micron. 33: 609-26. PMID: 12475558
- Theoretical and vibrational spectroscopic analysis of the CO stretching mode of cholesteryl alkanoates: the particular case of the cholesteryl acetate. | Zanoun, A., et al. 2005. Spectrochim Acta A Mol Biomol Spectrosc. 62: 547-51. PMID: 16099704
- Regio and stereoselective oxidations of unsaturated steroidal compounds with H2O2 mediated by CH3ReO3. | Ballistreri, FP., et al. 2006. Steroids. 71: 565-77. PMID: 16569418
- Prediction of solubility of drugs and other compounds in organic solvents. | Abraham, MH., et al. 2010. J Pharm Sci. 99: 1500-15. PMID: 19774653
- Crystal structure and thermal vibrations of cholesteryl acetate from neutron diffraction at 123 and 20 K. | Weber, HP., et al. 1991. Acta Crystallogr B. 47 (Pt 1): 116-27. PMID: 2025409
- The caveolin-binding motif of the pathogen-related yeast protein Pry1, a member of the CAP protein superfamily, is required for in vivo export of cholesteryl acetate. | Choudhary, V., et al. 2014. J Lipid Res. 55: 883-94. PMID: 24598142
- Synthesis of the 8,19-Epoxysteroid Eurysterol A. | Taspinar, Ö., et al. 2020. Chemistry. 26: 4256-4260. PMID: 32031278
- Two new halogenated sesquiterpene lactones from Palisada intermedia. | Doan, VT., et al. 2023. Nat Prod Res. 1-6. PMID: 36688708
- Properties of cholesteryl esters in pure and mixed monolayers. | Kwong, CN., et al. 1971. J Lipid Res. 12: 31-5. PMID: 5542702
- Cholesteryl esters on the body surfaces of the camel tick, Hyalomma dromedarii (Koch, 1844) and the brown dog tick, Rhipicephalus sanguineus (Latreille, 1806). | Sobbhy, H., et al. 1994. Exp Appl Acarol. 18: 265-80. PMID: 7628247
- Structures of nanoparticles prepared from oil-in-water emulsions. | Sjöström, B., et al. 1995. Pharm Res. 12: 39-48. PMID: 7724486
- Identification of thermal oxidation products of cholesteryl acetate. | Bortolomeazzi, R., et al. 1994. J Chromatogr A. 683: 75-85. PMID: 7952017
- Oral absorption efficiency of acid-labile antibiotics from lipid-drug delivery systems. | Patel, SP. and Jarowski, CI. 1975. J Pharm Sci. 64: 869-72. PMID: 807706
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Cholesteryl acetate, 25 g | sc-214693 | 25 g | $114.00 | |||
Cholesteryl acetate, 100 g | sc-214693A | 100 g | $317.00 |
