Ammonium hydrogencitrate (CAS 3012-65-5)
QUICK LINKS
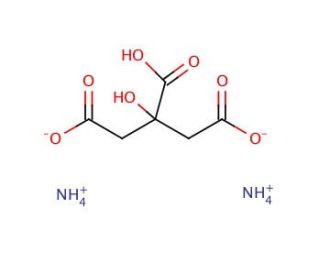
Ammonium hydrogencitrate, with the chemical formula C6H7O7NH4, is an organic ammonium salt derived from citric acid, where one hydrogen atom is replaced by an ammonium ion. This compound is highly valued in analytical and environmental chemistry for its chelating properties, allowing it to form stable complexes with metal ions. This ability makes it crucial in water treatment processes, where it aids in the removal of metallic contaminants by binding to metal ions and preventing their precipitation. In research contexts, ammonium hydrogencitrate is extensively used to study metal ion transport and absorption due to its effective chelation, which offers a controlled environment for examining metal behavior in various systems, including biological ones. Additionally, it finds application in biochemistry as a component of buffer solutions, maintaining pH stability during enzymatic reactions and other sensitive biochemical processes. Its utility is further extended to the field of nanotechnology, where it serves as a stabilizing agent in the synthesis of nanoparticles, helping to prevent aggregation and ensuring the production of uniformly dispersed nanoparticles. This makes ammonium hydrogencitrate a versatile agent in scientific research, contributing significantly to advancements in environmental management, biochemistry, and materials science.
Ammonium hydrogencitrate (CAS 3012-65-5) References
- Effect of additives on electrokinetic properties of colloidal alumina suspension. | Bhattacharjee, S., et al. 2002. J Colloid Interface Sci. 254: 95-100. PMID: 12702429
- Analytical procedure for the simultaneous voltammetric determination of toxic metals in dialysis fluids. | Locatelli, C. 2003. Anal Bioanal Chem. 376: 518-23. PMID: 12739100
- MALDI analysis of oligonucleotides directly from montmorillonite. | Zagorevskii, DV., et al. 2006. J Am Soc Mass Spectrom. 17: 1265-70. PMID: 16809045
- Retention of ionisable compounds on high-performance liquid chromatography XVII. Estimation of the pH variation of aqueous buffers with the change of the methanol fraction of the mobile phase. | Subirats, X., et al. 2007. J Chromatogr A. 1138: 203-15. PMID: 17118378
- Analytical procedure for the simultaneous voltammetric determination of trace metals in food and environmental matrices. Critical comparison with atomic absorption spectroscopic measurements. | Melucci, D., et al. 2007. Ann Chim. 97: 141-51. PMID: 17822223
- Matrix-assisted laser desorption ionization-time of flight mass spectrometry-based functional assay for rapid detection of resistance against β-lactam antibiotics. | Sparbier, K., et al. 2012. J Clin Microbiol. 50: 927-37. PMID: 22205812
- Optimized method for Acinetobacter species carbapenemase detection and identification by matrix-assisted laser desorption ionization-time of flight mass spectrometry. | Álvarez-Buylla, A., et al. 2013. J Clin Microbiol. 51: 1589-92. PMID: 23447631
- Effect of chemicals on fungal alpha-amylase activity. | Ali, FS. and Abdel-Moneim, AA. 1989. Zentralbl Mikrobiol. 144: 623-8. PMID: 2515680
- Bottom-up synthesis of nitrogen and oxygen co-decorated carbon quantum dots with enhanced DNA plasmid expression. | Yadegari, A., et al. 2019. Colloids Surf B Biointerfaces. 184: 110543. PMID: 31627102
- Synthesis of a novel nanocomposite containing chitosan as a three-dimensional printed wound dressing technique: Emphasis on gene expression. | Azadmanesh, F., et al. 2021. Biotechnol Prog. 37: e3132. PMID: 33527746
- Microbiological assay for organic compounds in seawater. I. Quantitative assay procedures and biotin distribution. | Litchfield, CD. and Hood, DW. 1965. Appl Microbiol. 13: 886-94. PMID: 5866037
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Ammonium hydrogencitrate, 100 g | sc-202938 | 100 g | $47.00 | |||
Ammonium hydrogencitrate, 500 g | sc-202938A | 500 g | $90.00 | |||
Ammonium hydrogencitrate, 2.5 kg | sc-202938B | 2.5 kg | $244.00 |
