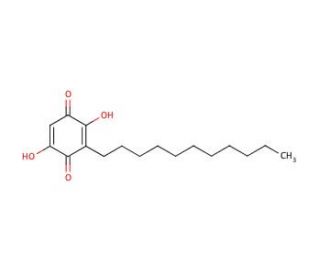
Embelin (CAS 550-24-3)
See product citations (5)
QUICK LINKS
Embelin is widely used in the field of biochemistry and molecular biology for its role as a modulator of various cellular processes. The compound is particularly recognized for its inhibition of the X-linked inhibitor of apoptosis protein (XIAP), which makes it valuable in the study of programmed cell death mechanisms. Researchers investigate the effects of embelin in understanding how apoptosis can be induced in cells, particularly within the context of cancer research, to elucidate potential strategies for cell regulation. In addition to its application in apoptosis studies, embelin′s antioxidant properties are leveraged in research examining the impact of oxidative stress on cellular health. The compound is also instrumental in probing the intricate signaling pathways implicated in inflammation and cell survival, providing insights into the complex network of intracellular communication.
Embelin (CAS 550-24-3) References
- Antibacterial activity of embelin. | Chitra, M., et al. 2003. Fitoterapia. 74: 401-3. PMID: 12781816
- Discovery of embelin as a cell-permeable, small-molecular weight inhibitor of XIAP through structure-based computational screening of a traditional herbal medicine three-dimensional structure database. | Nikolovska-Coleska, Z., et al. 2004. J Med Chem. 47: 2430-40. PMID: 15115387
- Free radical scavenging reactions and antioxidant activity of embelin: biochemical and pulse radiolytic studies. | Joshi, R., et al. 2007. Chem Biol Interact. 167: 125-34. PMID: 17379198
- Embelin inhibits pancreatic cancer progression by directly inducing cancer cell apoptosis and indirectly restricting IL-6 associated inflammatory and immune suppressive cells. | Peng, M., et al. 2014. Cancer Lett. 354: 407-16. PMID: 25128650
- Embelin, a small molecule quinone with a co-clinical power for castrate-resistant prostate cancer. | Poojari, RJ. 2014. Front Pharmacol. 5: 184. PMID: 25152733
- Embelin (2,5-dihydroxy-3-undecyl-p-benzoquinone): a bioactive molecule isolated from Embelia ribes as an effective photodynamic therapeutic candidate against tumor in vivo. | Joy, B., et al. 2014. Phytomedicine. 21: 1292-7. PMID: 25172792
- Synthesis and anti-proliferative activities of new derivatives of embelin. | Singh, B., et al. 2014. Bioorg Med Chem Lett. 24: 4865-70. PMID: 25240254
- The XIAP inhibitor Embelin enhances TRAIL-induced apoptosis in human leukemia cells by DR4 and DR5 upregulation. | Hu, R., et al. 2015. Tumour Biol. 36: 769-77. PMID: 25293521
- Embelin inhibits TNF-α converting enzyme and cancer cell metastasis: molecular dynamics and experimental evidence. | Dhanjal, JK., et al. 2014. BMC Cancer. 14: 775. PMID: 25336399
- Embelin sensitizes acute myeloid leukemia cells to TRAIL through XIAP inhibition and NF-κB inactivation. | Yang, T., et al. 2015. Cell Biochem Biophys. 71: 291-7. PMID: 25358405
- Embelin (2,5-Dihydroxy-3-undecyl-p-benzoquinone) for photodynamic therapy: study of their cytotoxicity in cancer cells. | Joy, B., et al. 2015. Appl Biochem Biotechnol. 175: 1069-79. PMID: 25359675
- Inhibition of Angiogenesis and Nitric Oxide Synthase (NOS), by Embelin & Vilangin Using in vitro, in vivo & in Silico Studies. | Narayanaswamy, R., et al. 2014. Adv Pharm Bull. 4: 543-8. PMID: 25671187
- Embelin Reduces Systemic Inflammation and Ameliorates Organ Injuries in Septic Rats Through Downregulating STAT3 and NF-κB Pathways. | Zhou, XL., et al. 2015. Inflammation. 38: 1556-62. PMID: 25682469
- Antitumor, anti-inflammatory and analgesic property of embelin, a plant product. | Chitra, M., et al. 1994. Chemotherapy. 40: 109-13. PMID: 7510605
Ordering Information
| Product Name | Catalog # | UNIT | Price | Qty | FAVORITES | |
Embelin, 10 mg | sc-201555 | 10 mg | $89.00 | |||
Embelin, 50 mg | sc-201555A | 50 mg | $339.00 |
